Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Newly Discovered Molecule Cuts Bacterial Proteins Short
Drug Discovery: Orthoformimycin, isolated from a microbial culture, shows novel antibiotic activity
by Erika Gebel Berg
August 8, 2013

The world’s vast diversity of microorganisms produces a wide array of chemicals, many as yet undiscovered. Some of this molecular bounty can provide leads for new drugs. Now, a search of the chemical output from thousands of microbes has revealed a compound with an unusual structure that could form the basis of a new class of antibiotics (ACS Chem. Biol. 2013, DOI: 10.1021/cb4004095).
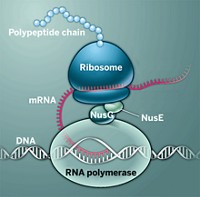
Scientists have been hunting for the next molecular weapon against the increasing ranks of antibiotic-resistant bacteria. Because many known antibiotics arrest bacterial protein synthesis by binding to ribosomes—the cellular machines that connect amino acids—identifying compounds with such activity is a tried-and-true method for seeking new drugs.
Claudio O. Gualerzi of the University of Camerino, in Italy, and his team screened extracts from the cultures of 5,000 species of bacteria and fungi. The researchers added each of these broths to two cell-free protein expression systems—one that used Escherichia coli components and another that used yeast machinery. One microorganism—a close relative of the bacterium Streptomyces griseus—produced an extract that shut down protein expression in the E. coli system but not the yeast system. Blocking protein synthesis in prokaryotes but not in eukaryotes bodes well for a compound that could be used in humans.
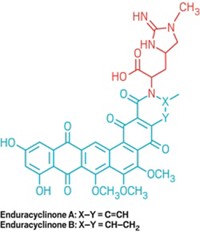
To identify the molecule responsible, the researchers then separated the Streptomyces extract with high-performance liquid chromatography, checking each fraction for its ability to block protein synthesis in the E. coli expression system. With additional separations, they isolated the active compound and determined the structure using nuclear magnetic resonance spectroscopy. The compound included a collage of functional groups, but its unique feature was an orthoformate moiety—a trio of oxygen atoms attached to a central carbon. So Gualerzi and his team named the molecule orthoformimycin.
Next, the researchers evaluated orthoformimycin’s ability to block protein synthesis. In the E. coli expression system, the molecule inhibited more than 80% of the translation of a range of messenger RNA templates. Furthermore, the researchers showed that the compound binds with the 50S subunit of the E. coli ribosome, altering the coordinated movements of mRNA and transfer RNA. This disruption halts the addition of amino acids to the peptide chain, thus stopping translation through a novel mechanism.
Although the studies in the cell-free system pointed to orthoformimycin as a potent antibacterial weapon, the story changed when the researchers added the compound to cell cultures. Orthoformimycin has poor antimicrobial activity, and Gualerzi suspects that intact cells can either block the molecule’s entry or rapidly eject it. He’s working to crystallize orthoformimycin in complex with the 50S subunit, to see which parts of the molecule are important for binding and which parts can be tweaked to improve its transport into bacteria.
Though it’s unclear whether this molecule will lead to new antibiotics, it could influence how researchers think about the problem, says Douglas B. Weibel of the University of Wisconsin, Madison. “This compound—and its derivatives—may more importantly shed new insight into translation and reset our imagination for how to inhibit this cellular machinery in bacteria.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter