Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Nanobiopsies On Living Cells
Cell Biology: A robotic nanopipette gently removes femtoliter samples from single cells without killing them
by Louisa Dalton
December 6, 2013

When scientists investigate the inner contents of a cell, the methods they use often end up killing it. But now bioengineers introduce a kind of cellular biopsy—a simple system for gently extracting femtoliters of material from a cell without destroying it (ACS Nano 2013, DOI: 10.1021/nn405097u). The technique permits repeat sampling of a living cell—a powerful tool for studying a cell’s dynamic biochemistry.

Much of what cell biologists know comes from studying populations of cells. Yet “no two cells are alike,” says Nader Pourmand, a biomolecular engineer at the University of California, Santa Cruz. To really understand the range of chemistries going on inside cells, it’s important to analyze single cells—and to do that over time, he says. For example, a cancer cell’s biochemistry differs greatly from a healthy one, and it changes dramatically as the cell moves from a precancerous stage to malignancy. Scientists would like to take samples repeatedly from the same cell to trace that evolution.
Current methods that use micropipettes or microdissection to get at a cell’s contents are generally fatal to the cell. A few groups have found less harmful ways to specifically remove RNA from a cell. Yet a method for general extraction from a live cell has remained elusive, Pourmand says.
Pourmand and his group were already using glass pipettes with 100-nm-sized openings to detect certain biomolecules and ions. So they decided to design a system that would guide the nanopipette like a syringe to pump material in and out of single cells.
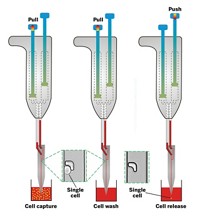
The team customized a scanning ion conductance microscope (SICM) to gently impale the cells they want to study. First, the researchers roughly align a nanopipette filled with an organic solvent over living cells in cell culture media. Both the media and the liquid in the pipette contain charged electrolytes, which allow a current to flow between the nanopipette tip and the cells. Then the researchers push a button, and the SICM computer takes over, guiding the nanopipette toward the cells. The computer can sense where a single cell sits because the current decreases as the pipette tip gets closer to the cell’s surface. Just before the pipette touches the cell, it stops, then quickly plunges down 1 μm to pierce the cell membrane.
Once in, the computer applies a negative voltage across the tip of the pipette. Through a phenomenon called electrowetting, the voltage alters the surface tension between the organic solution in the pipette and the watery solution of the cell. As a result, the pipette sucks up about 50 femtoliters, about 1% of a human cell’s volume.
Pourmand was happily surprised to see that, after sampling, the cells maintain their shape and membrane integrity, even after 10 punctures.
To show that their method could extract useful material, they plucked out and sequenced intracellular messenger RNA from human cancer cells and mitochondrial DNA from human fibroblasts. They could target mitochondria by labeling them with a fluorescent dye and placing the nanopipette above an area rich with the organelles.
Pourmand’s group has many further plans for the technology. Already, they have created a single, multipurpose nanopipette tip that can squirt material into a cell, suck samples out of it, and sense what’s in the sample. And they are creating techniques for doing single-cell metabolic analysis by sampling not just nucleic acids, but proteins, sugars and other biomolecules.
“You can extract anything. That is the beauty of it,” says Yuri Korchev, professor of biophysics at Imperial College London. Such flexibility, he says, permits many different types of measurements, such as single-cell diagnostic tests or drug screening. “It has powerful potential,” he says. “I see a hundred possibilities.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter