Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Peptides Self-Assemble To Form Catalytic Amyloids
Findings have implications for origins of life and disease-related amyloid toxicity
by Celia Henry Arnaud
March 17, 2014
| A version of this story appeared in
Volume 92, Issue 11
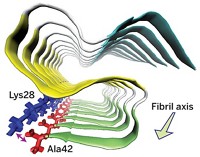
A new study supports the hypothesis that the earliest enzymes may have been self-assembling peptides instead of full-length proteins. Ivan V. Korendovych of Syracuse University; William F. DeGrado of the University of California, San Francisco; and coworkers designed short peptides that self-assemble into catalytic amyloids (Nat. Chem. 2014, DOI: 10.1038/nchem.1894). Similar peptide aggregates are a hallmark of diseases such as Alzheimer’s. In this case, the amyloids are made of seven-residue peptides consisting of alternating apolar and polar residues. Leucines provide the hydrophobicity needed to drive amyloid formation; two histidines bind zinc ions, which stabilize the amyloid structure and allow it to catalyze acyl ester hydrolysis. “The study strengthens the hypothesis that amyloids played an important role in the origin of life,” says Roland Riek, a chemist at ETH Zurich who is studying functional amyloids and establishing amyloid structure-function relationships. In addition, says David S. Eisenberg, a chemist at UCLA who studies amyloid structure and formation, the work adds support to one of the hypotheses for the toxic effects of some amyloid fibers: that such fibers bind metals and as a result produce harmful reactive oxygen species.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter