Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Theoretical Chemistry
Iridium Dressed To The Nines
Periodic Table: IrO4+ is the first molecule with an element in the +9 oxidation state
by Stephen K. Ritter
October 23, 2014
| A version of this story appeared in
Volume 92, Issue 43

The oxidation state of an atom in a molecule is an important means of electron bookkeeping and determining structure and bonding, so the question of just how high oxidation states can go has long piqued chemists’ interest. That ceiling has now reached what was once believed to be an impossibly high level with the preparation of IrO4+, the first example of a molecule with an oxidation state of +9.
“Wow. The discovery that chemical compounds in the +9 oxidation state may be stable enough to isolate is provocative, to say the least,” says Gregory S. Girolami, an inorganic chemist at the University of Illinois, Urbana-Champaign. “If confirmed, the iridium compound would be a worthy member of the elite list of chemical species, such as noble-gas compounds, that exist despite once being thought permanently ‘nonexistable.’ ”
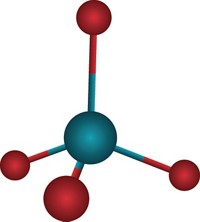
The highest observed oxidation state had been +8, which occurs in only a few tetroxide compounds—RuO4, OsO4, IrO4, and XeO4. These are molecules in which the central atom has plenty of valence electrons to give away and the high charge can be stabilized by small, highly electronegative ligands such as fluorine or oxygen. Among the +8 molecules, IrO4 stands out because the central iridium atom still has one more 5d valence electron to give.

Researchers led by Sebastian Riedel of Albert Ludwigs University, in Freiburg, Germany; Mingfei Zhou of Fudan University, in Shanghai; Jun Li of Tsinghua University, in Beijing; and Gary J. Schrobilgen of McMaster University, in Hamilton, Ontario, have now coaxed iridium to go all the way (Nature 2014, DOI: 10.1038/nature13795).
To make IrO4+, Zhou’s group blasted an iridium metal target with a pulsed laser in an argon atmosphere spiked with O2. The researchers studied the reaction products by mass spectrometry and infrared photodissociation spectroscopy. By coupling experimental work by Zhou’s group and computational modeling by Riedel’s and Li’s groups, the researchers determined that the most stable IrO4+ isomer has tetrahedral geometry and four terminal Ir=O bonds.
Schrobilgen’s group attempted to synthesize an isolable IrO4+ salt by treating iridium oxides with strong oxidizing reagents such as O2SbF6 and XeF6. The researchers haven’t found the right combination to make isolable IrO4+ so far, but they haven’t given up trying.
“New well-documented oxidation states are rarer than new elements,” comments theoretical chemist Pekka Pyykkö of the University of Helsinki, in Finland. Pyykkö has studied iridium’s propensity for taking on many oxidation states. The Ir +9 compound extends the list so that all positive oxidation states from Ir +1 to Ir +9 are now known, Pyykkö notes, along with a few negative ones.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter