Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
How To Build A Biobased Copolymer
Sustainability: Chemists can now make cost-competitive polyesters entirely from biobased monomers
by Stephen K. Ritter
May 29, 2014

By combining biosynthesis expertise with polymer synthesis and engineering skills, a University of Minnesota research team has laid the groundwork for producing copolymers that have a wide range of properties and applications and are made entirely from sugar-derived building blocks. The new materials are the first biobased block copolymers with the potential to go stride-for-stride in cost and performance with petroleum-based polyester elastomers and hard plastics.
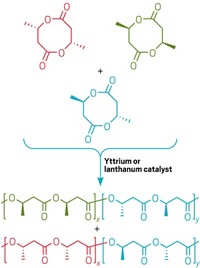
The development hinged on chemical engineering and materials science professor Kechun Zhang and postdoctoral associate Mingyong Xiong first designing a biosynthetic route to make the six-membered ring β-methyl-δ-valerolactone (MVL) as a new biobased monomer. Chemistry professor Marc A. Hillmyer and graduate student Deborah K. Schneiderman, in collaboration with chemical engineering and materials science professor Frank S. Bates, then controllably copolymerized MVL with lactide, a biobased cyclic diester already used as a biodegradable polymer building block (Proc. Natl. Acad. Sci. USA 2014, DOI: 10.1073/pnas.1404596111).
“The production of these block copolymers is a comprehensive feat that has accomplished several important aspects needed for the development of sustainable materials with tunable properties that are capable of competing with petroleum-derived engineering plastics,” comments Texas A&M polymer chemist Karen L. Wooley.
The most successful biobased synthetic polymer to date is poly(lactide), or PLA, a rigid aliphatic polyester used in applications including compostable drinking cups and in medical devices. However, the brittle nature of PLA and other commercial biobased polyesters has thwarted their broad use.

Polymer chemists have been trying to make PLA block copolymers with improved properties, Hillmyer explains, but the copolymer blocks are derived from petroleum or from prohibitively expensive renewable sources. And affordable biobased compounds such as the five-membered ring γ–valerolactone aren’t amenable to easy polymerization. The Minnesota team went to the drawing board and designed MVL to have just the right properties to work with lactide.
To synthesize MVL, the researchers built on a metabolic pathway previously engineered into yeast and bacteria for synthesizing mevalonate, an intermediate on the way to making terpenoids and the antimalarial drug artemisinin. They added fungal enzymes to the pathway to expand it to prepare MVL directly from glucose. The yields suggest MVL can be commercially produced for $2.00 per kg—a number that gives it economic traction, Hillmyer says.
MVL forms an amorphous, rubbery polymer on its own. But adding lactide to the polymerization in progress leads to a more rigid, crystalline polymer. By controlling the ratio of the two polymer blocks, the Minnesota team can tune the mechanical properties from those of soft, stretchable elastomers that could be used in the soles of running shoes to the stiff and tough properties of thermoplastics appropriate for building automobiles.
“Many approaches have laudably addressed the two main concerns that sustainable polymers have a green birth and a green death, but the vast majority fall short with respect to the most important polymeric property—cost,” observes polymer chemist Stephen A. Miller of the University of Florida. “This new approach not only achieves the economic viability threshold but also delivers a novel class of green copolymers with properties already sought by the plastics industry.”





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter