Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
New Polymer Nanocapsules Come With Built-In Channels
Nanomaterials: The particles’ nanosized pores open in response to high pH, releasing their cargo
by Prachi Patel
August 19, 2014

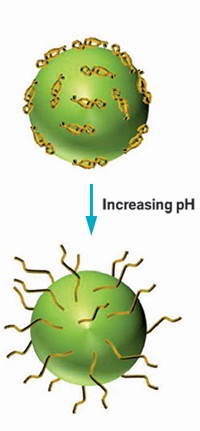
Researchers have made nanosized polymer capsules with built-in channels that release the particles’ payload when triggered by a high pH (Macromolecules 2014, DOI: 10.1021/ma501099f). The nanocapsules could be useful for the controlled delivery of drug molecules and proteins of specific sizes inside the body.
Polymer capsules have become a popular tool in the pharmaceutical and personal care industries to deliver drugs, hormones, and fragrances under certain conditions. Researchers have typically used linear block copolymers to form vesicles that swell or become porous when exposed to heat or certain wavelengths of light, releasing their contents.
Chemists Heng Hu and Guojun Liu at Queen’s University, in Ontario, wanted to make capsules with pores built right in. These channels could give researchers control over the size of molecular cargo that enters and leaves the hollow particles. Previous copolymer capsules have been micrometers wide. The duo wanted to make nanometer-wide ones for possible drug delivery applications. At that size, the particles could easily permeate the digestive tract and blood vessels, Hu says.
To make the new capsules, the chemists picked a miktoarm star copolymer, a class of star-shaped polymers in which the arms have different chemical compositions. The researchers chose three polymer arms: a poly(tert-butyl acrylate) (PtBA) chain, a poly(2-cinnamoyloxyethyl methacrylate) (PCEMA) chain, and a poly(ethylene oxide) (PEO) chain.
After synthesizing the copolymer, they slowly added water to a solution of the material in tetrahydrofuran. The copolymer self-assembled into 200-nm-wide vesicles with walls made of PtBA and PCEMA. The PEO chains helped stabilize the vesicles in water, and the PtBA chains formed cylinders within the PCEMA.
The researchers next cross-linked the PCEMA chains with ultraviolet light, and then treated the vesicles with trifluoroacetic acid to hydrolyze the tert-butyl group on the PtBA chains, exposing acrylic acid groups. At an acidic pH, the now poly(acrylic acid) chains blocked the entrances of the capsules’ channels. But at a basic pH, the chains moved out of the way, allowing access to the channels.
Using atomic force and transmission electron microscopy, the chemists found that the particles were hollow and round and that the channels were 9 nm wide. To test the particles, Hu and Liu packed the hollow globules with PEO chains tagged with pyrene butyric acid, a fluorescent dye, and placed them in an aqueous solution with a pH of 2.85. When they increased the pH to 11.23, the fluorescent intensity of the solution surged, showing that the capsules had unloaded their cargo.
Hu says that making the capsules is easy and requires only simple equipment. He thinks they can optimize the process to shorten the three-day preparation time. The researchers are now trying to tweak the chemistry to make the capsules biocompatible and biodegradable.
Ian Manners, a chemist at the University of Bristol, in England, calls this an elegant design and an important advance. While drug-release is an obvious application, he says, the new work represents “a small but significant step towards the design of artificial nanostructures that exhibit some features of cell-like behavior.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter