Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Two-Pronged Peptide Fingerprinting With UV And Mass Spec
Analytical Techniques: Coupling UV spectroscopy with high-resolution mass spectrometry yields structurally specific fingerprints
by Celia Henry Arnaud
April 20, 2015
| A version of this story appeared in
Volume 93, Issue 16
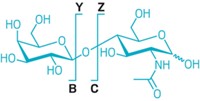
High-resolution mass spectrometry is a wonder for identifying similar molecules in a mixture, but users of the technique still have a hard time distinguishing isomers. Oleg V. Boyarkin and Vladimir Kopysov of ETH Lausanne and Alexander Makarov of Thermo Fisher Scientific have found a way to improve structural identification of isomeric biomolecules by combining ultraviolet spectroscopy with high-resolution mass spec (Anal. Chem. 2015, DOI: 10.1021/acs.analchem.5b00822). They use UV laser pulses to break apart cryogenically cooled ions and measure the mass spectrum of the resulting fragments with an Orbitrap mass spectrometer. The two-dimensional spectra provide structurally specific fingerprints, which the researchers used to distinguish peptide stereoisomers and peptides that differ only in the site of phosphorylation. They were even able to quantitate the relative composition of mixtures. “The fundamental science is very nice, but I do not envision this becoming a practical analytical method,” says James P. Reilly of Indiana University. Few peptide residues absorb UV light, and high-resolution UV spectra of various peptides are not predictable, Reilly notes. For most labs, the combination of a liquid helium-cooled ion storage system, an Orbitrap mass spectrometer, and a tunable UV laser would be prohibitively expensive, he says. But practicality, he adds, “is not everything.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter