Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Adding Pep To PEPPSI
Cross-Coupling: Chemists develop a better catalyst for adding secondary alkyl groups to five-membered heterocycles
by Stephen K. Ritter
July 6, 2015
| A version of this story appeared in
Volume 93, Issue 27
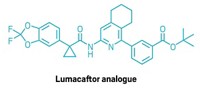
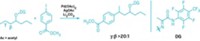
The secret to the palladium-catalyzed cross-coupling reactions is often the judicious choice of catalyst ligand. Michael G. Organ’s group at York University, in Toronto, in collaboration with researchers at Dow Chemical and Eli Lilly & Co., have revealed a bit more of that secret by learning how to tweak the structure of a pyridine-N-heterocyclic-carbene-based ligand system called PEPPSI to carry out selective Negishi couplings of secondary alkyl groups with five-membered heterocyclic rings, a reaction not possible before (Angew. Chem. Int. Ed. 2015, DOI: 10.1002/anie.201503941). Negishi coupling works for adding branched alkyl groups to six-membered rings, but attempts to do the same with five-membered rings gave a mixture of linear and branched isomers. Organ’s group found that adjusting the structure of a commercial PEPPSI ligand they previously developed, by switching from isopentyl to isoheptyl substituents and adding chlorines, increases the bulk and alters the electronic properties to favor the desired branched isomers.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter