Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Radical Polymer Approach
Polymerization: Organic initiators replace metal catalysts
by Bethany Halford
January 15, 2015
| A version of this story appeared in
Volume 93, Issue 3

A protocol for ring-opening metathesis polymerization, or ROMP, that uses organic initiators rather than transition-metal catalysts has been developed. The new polymerization method could be a boon to makers of polymers with electronic or biological applications.
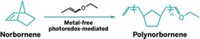
Today, polymer producers use ROMP to make such materials as polynorbornene, trade named Norsorex, which can be used as a shock absorber and a high-grip material, as well as for water filtration. Polydicyclopentadiene, which is used to make body parts for tractors and construction equipment, is also made via ROMP.
But polymer makers have been limited in their ability to expand their offerings with ROMP because traces of the transition metals used to initiate polymerization can be left behind in the product. That’s a problem in electronic applications, where the residual metals affect the polymer’s conductivity and capacitance. It’s also troublesome for polymers with biological applications because the metals used for ROMP are toxic.
With these limitations in mind, University of Washington, Seattle, chemists Andrew J. Boydston, Kelli A. Ogawa, and Adam E. Goetz set out to initiate a ROMP reaction without metals. Instead, they used radical cations made from one-electron oxidation of electron-rich vinyl ethers (J. Am. Chem. Soc. 2015, DOI: 10.1021/ja512073m).
The researchers think the radical cation reacts with a double bond to produce a cyclobutane radical cation. If the double bond is part of a small ring system, such as norbornene, the ring’s strain will prompt the cyclobutane radical cation to open up, initiating the polymerization process. Trial and error led the chemists to photoredox chemistry as the best method for creating the radical cation initiator.
So far, the researchers have used only norbornene as a monomer in metal-free ROMP, but Boydston tells C&EN they want to expand the scope of the reaction. “Norbornene is too limited,” he says. “One of the reasons that metal-mediated ROMP is so successful is because it works on almost anything.”
“If this discovery can be applied to a broad array of strained cyclic olefins, the development of metal-free approaches to various established and new materials would be quite exciting,” comments Marc A. Hillmyer, a polymer expert at the University of Minnesota.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter