Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Dormant Virus Can Wake Up In Some Patients With Lou Gehrig’s Disease
Neuroscience: Retrovirus sitting in “junk DNA” of human genome could contribute to neurodegeneration seen in ALS
by Michael Torrice
September 30, 2015
| A version of this story appeared in
Volume 93, Issue 39

Our chromosomes hold a partial record of prehistoric viral infections: About 8% of our genomes come from DNA that viruses incorporated into the cells of our ancestors. Over many millennia, these viral genes have accumulated mutations rendering them mostly dormant.
But one of these viruses can reawaken in some patients with amyotrophic lateral sclerosis (ALS), a progressive muscle wasting disease commonly known as Lou Gehrig’s disease. A new study demonstrates that this so-called endogenous retrovirus can damage neurons, possibly contributing to the neurodegeneration seen in the disease.
The findings raise the possibility that antiretroviral drugs, similar to those used to treat HIV, could slow the progression of ALS in some patients.
Avindra Nath, the study’s lead author and the clinical director at the National Institute of Neurological Disorders & Stroke, typically studies HIV and its effects on the brain. But an HIV patient he treated in 2006 led him to start investigating ALS.
In addition to being HIV positive, this patient also had an ALS-like disorder. When Nath put him on a typical cocktail of antiretroviral drugs to treat the HIV infection, something odd happened—his ALS symptoms got better. “I thought this was really bizarre,” Nath says. “If you have ALS, the mortality rate is almost 100%, there is no way ALS gets better.”
When Nath started to read through the medical literature on ALS, he found a handful of similar cases. He also noticed studies reporting that enzymes called reverse transcriptases were active in some ALS patients. These enzymes are a marker for infection by retroviruses, such as HIV, because they allow the viruses to incorporate their genetic code into our cell’s DNA. ALS researchers had been looking for a possible retrovirus responsible for this reverse transcriptase activity, but had yet to find one.
Nath wondered if the culprit wasn’t an invading virus, but one lying dormant in the patients’ own genomes. In 2011, he and his colleagues reported that brain samples from deceased ALS patients had elevated levels of RNAs corresponding to the RNA polymerase of a dormant virus called human endogenous retrovirus-K (HERV-K).
Now the researchers show that RNA from the virus’s other two genes are also elevated in brain samples from ALS patients compared to healthy people and Alzheimer’s disease patients (Sci. Transl. Med. 2015, DOI: 10.1126/scitranslmed.aac8201).
In the new study, the team also tried to determine whether HERV-K could contribute to the pathology of the disease. They first used gene editing techniques to activate the dormant virus in cultures of human neurons. Waking up HERV-K caused the neurons to shrink and die.
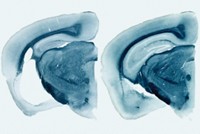
In another experiment, the researchers genetically engineered mice to express one of HERV-K’s proteins only in neurons. The regions of these animal’s brains that control motor functions were smaller than those in normal mice, suggesting that the viral protein had destroyed neurons. These mice also displayed behaviors indicative of this neurodegeneration, including spastic movements and muscle weakness.
“A key question is whether the increased expression of HERV-K in the brains of ALS patients is a cause or a consequence of the disease,” says Jeremy A. Garson, a virologist who has studied the possible retrovirus-ALS connection at University College London. The new data “have gone some way to answering this by demonstrating not only that elevated HERV-K expression can cause neurotoxicity but also that the elevated expression is not just a nonspecific consequence of neuronal injury.”
The animal results, in particular, are exciting because the mice display some pathological signatures seen in ALS patients, says Stephen N. Scelsa, a neurologist who also has studied retroviruses and ALS at Mount Sinai Hospital, in New York. He thinks the researchers need to look at larger numbers of ALS patient samples to confirm and determine the prevalence of HERV-K activation in the disease.
“But if the virus is important in triggering the disease, then it would certainly be easier to target a viral infection of the nervous system than it is to target a neurodegenerative process that we still don’t fully understand,” he says.
Nath’s team is planning a short clinical study to investigate the effect of a cocktail of anti-HIV drugs on levels of HERV-K in ALS patients.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter