Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
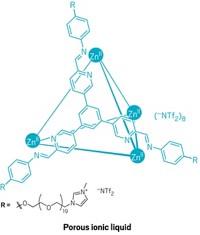
Materials
This Liquid Has Holes In It, Thanks To Chemistry
Materials Science: Liquid made with cage compounds has permanent pores and could one day help with gas separations
by Stu Borman
November 12, 2015
| A version of this story appeared in
Volume 93, Issue 45

How is a new type of liquid like the London Underground? In both, one must mind the gaps.
Stuart James of Queen’s University Belfast and coworkers have devised a liquid with permanent porosity (Nature 2015, DOI: 10.1038/nature16072). Such holey fluids could be useful in gas separation, process chemistry, and other applications, if they can be made economically. Their persistent porosity comes from hollow organic cage molecules coated with solvent-soluble surface groups. The cage openings are too small to be clogged by the surface groups or by large solvent molecules in the surrounding solvent.
Liquids contain spaces between their molecules, but they are tiny. Bubbles can be blown into liquids, but they will quickly float to the surface and dissipate—air bubbles in glass being a rare exception.
On the other hand, solids such as zeolites and metal-organic frameworks have permanent pores. They can be used to separate molecules by size and can host added catalysts to drive chemical reactions. But unlike liquids, these materials can’t flow through channels or be smoothed onto surfaces.
James got the idea for holey liquids a decade ago, when a colleague, Queen’s University chemical engineer David W. Rooney, wondered whether mixtures of porous solids and liquids might be pumped through pipes and used in continuous processes more easily than is generally possible with porous solids alone.
Porous solids expert Russell Morris of the University of St. Andrews comments that the concept of liquids with permanent porosity has actually been around for decades, but “the practicalities of designing such a system take skill and perseverance. This work delivers real liquids that show that property, which is a great achievement.”
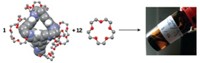
The researchers initially achieved liquid porosity by dissolving a hollow organic core structure coated with crown-ether surface groups in a crown-ether solvent. To optimize gas absorption capacity, the team packed the fluid with as many cage molecules as possible—one for every 12 solvent molecules. When exposed to methane, the liquid absorbs eight times as much gas as the solvent alone.
But the crown ethers were hard to synthesize and the liquid thick and slow-flowing. So collaborators Andrew I. Cooper and Rebecca L. Greenaway at the University of Liverpool developed another porous liquid by coating a hollow organic cage with a mixture of diamines and dissolving it in the solvent hexachloropropene. The resulting porous liquid flows ten times as readily as the crown-ether-based material. The researchers synthesize the coated cages in a single step, and the solvent is available commercially.
In earlier work, Shannon M. Mahurin and Sheng Dai at Oak Ridge National Laboratory and coworkers created hollow colloidal silica nanoparticles with a fluidlike polymer surface coating (Angew. Chem. Int. Ed. 2015, DOI: 10.1002/anie.201409420), but the new porous liquids are easier to molecularly modify. Dai comments that “the liquids will open up new frontiers in how we think about porosity.”
The surface area and gas uptake of the new materials are too modest for them to compete with porous solids immediately, says cage compound specialist Michael Mastalerz of Germany’s Heidelberg University in a Nature commentary. “They should instead be seen as a prototype of a new class of material [that] will undoubtedly find technological applications” if these properties can be improved.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter