Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Water Affects Contact At Hydrophobic Interfaces
Direct measurements contradict indirect ones and theory, suggesting reevaluation of energetics in protein folding and other common processes
by Mitch Jacoby
March 2, 2015
| A version of this story appeared in
Volume 93, Issue 9
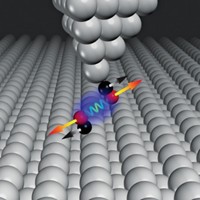
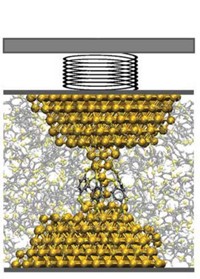
Energetics controlling the way two hydrophobic surfaces make contact and slide past one another play a central role in protein folding, lock-and-key type enzyme reactions, and other processes influenced by forces of adhesion and friction. Theoretical studies have shown that as hydrophobic surfaces come in contact even underwater, water is excluded from the interface, leading to dry contact. But until now, that scenario has not been probed directly by experiment. University of Akron polymer scientists Adrian P. Defante and Ali Dhinojwala and coworkers used a surface-sensitive spectroscopy method to directly examine the contact interface between hydrophobic polydimethylsiloxane (PDMS) and films of two materials regarded as equally hydrophobic—octadecyltrichlorosilane (OTS) and poly(vinyl-n-octadecyl carbamate) (PVNODC). In the absence of water, OTS and PVNODC respond identically to contact with PDMS. In water, however, PVNODC’s hydrophobic side chains rearrange and trap water at the interface, lowering the adhesion energy by 50% compared with the OTS-PDMS interface (Langmuir 2015, DOI: 10.1021/la504564w). Standard mathematical models used for calculating adhesion thermodynamics fail in this case, highlighting the need to better understand interfacial contact phenomena.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter