Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Carbon-Nanotube Electrodes Make Better Brain Connections
Neuroscience: Nanotube electrodes can stimulate and record from neurons without damaging brain tissue
by Katherine Bourzac
April 15, 2015

Electrodes made from carbon-nanotube fibers can both stimulate the brain and receive signals from neurons, which may improve prosthetic devices and brain stimulation therapies for neural disorders (ACS Nano 2015, DOI: 10.1021/acsnano.5b01060). Compared to conventional metal electrodes, the nanotube fibers cause less damage and make better electrical connections with brain tissue in rats.
To treat movement disorders such as Parkinson’s disease, doctors can implant metal electrodes in a patient’s brain and apply current, a therapy called deep-brain stimulation (DBS). Doctors determine the right frequency of stimulation through trial and error: Patients return to the clinic, report their symptoms, and receive electrode adjustments. Better electrodes that could not only stimulate but also record brain activity would lead to DBS devices that adjust the frequency of stimulation on the fly. These electrodes could also support more sophisticated body-computer interfaces, such as prosthetic limbs that better integrate with the peripheral nervous system.
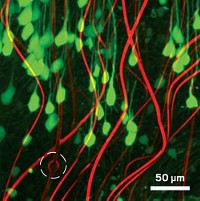
Matteo Pasquali, a chemist at Rice University, and his colleagues had been developing ways to make strong, conductive carbon-nanotube fibers. After a conversation with a neuroscientist, he realized that the fibers might make good electrodes for probing the brain. The fibers can be as small as 8 μm in diameter, yet retain good electrical conductivity, making them ideal for both delivering therapeutic stimulation and picking up faint signals from neurons.
Pasquali collaborated with Rice neural engineer Caleb Kemere to test 12-μm-diameter nanotube fibers in a rat model of Parkinson’s and compared them to conventional platinum electrodes coated with iridium. They implanted the electrodes in rat brains and used them to perform DBS. The impedance of the nanotube fibers—their tendency to resist carrying charge into the tissue—was about one-twentieth that of conventional electrodes. That allowed the nanotube electrodes—which are much smaller and thus capable of targeting brain regions more precisely—to deliver current as effectively as the larger metal electrodes.
What surprised Pasquali is the biocompatibility of the fibers, which he and his group had originally been developing for aerospace applications. In addition, Pasquali says, “we never talked about flexibility, but that may end up being the most important property of these fibers.” Their soft, silky feel make them a good mechanical match for the brain. Six weeks after implanting the electrodes in the rats, the researchers examined the tissue around the electrodes. Unlike the metal electrodes, carbon-nanotube implants did not trigger inflammation in the rat brains.
Nicholas A. Kotov, a materials scientist at the University of Michigan who is also developing nanomaterial coatings for electrodes, calls the results impressive. He adds the electrodes would work even better if they were thinner, and possibly if they were coated, which would lower their impedance further and improve their connection with brain tissue. Pasquali’s group is now designing multichannel arrays of the electrodes—groups of the devices that will interact with multiple areas in the brain.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter