Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Making Light Of Oxidative Stress In Cells
Analytical Chemistry: Researchers synthesize a fluorescent molecule that could clarify the connection between oxidative stress and disease
by Erika Gebel Berg
August 10, 2015
To unravel the biology behind diabetes, heart disease, cancer, and a host of other ailments, scientists may need a stress detector. Researchers have now developed a fluorescent probe for a key biomarker of oxidative stress, malondialdehyde, that may help scientists better understand the potential connection between an excess of reactive oxygen species and human disease (Anal. Chem. 2015, DOI: 10.1021/acs.analchem.5b02032). By visualizing oxidative stress in living cells, or perhaps someday in animals, scientists could monitor the efficacy of experimental treatments that might counter this cellular damage.

Oxidative stress occurs when reactive oxygen species—formed as a natural by-product of oxygen metabolism—overwhelm antioxidant defenses in the body. However, little is known about the situations that prompt oxidative stress in particular tissues. These radicals can violently react with cellular components, such as the lipids in membranes. When reactive oxygen species attack polyunsaturated fatty acids, they produce malondialdehyde. The most common method for detecting this biomarker is an assay that requires cell-killing conditions: a strong acid and high temperatures. Other detection methods—such as Raman spectroscopy, electrophoresis, and liquid chromatography—are also incompatible with live cells. Microscopy is a popular way to study living cells, so Zhihong Liu of Wuhan University, in China, and colleagues wanted to create a fluorescent probe that they could use to track malondialdehyde.
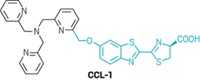
As a starting point, the researchers studied the reaction of malondialdehyde with benzohydrazide, which reacts readily with aldehydes. They tinkered with the structure of benzohydrazide so that it would react selectively with malondialdehyde, and then linked the compound to a fluorophore, 1,8-naphthalimide. The end result was a fluorescent molecule, named MDAP-1, whose glow increased 170-fold upon reacting with malondialdehyde.
The researchers then checked to see if MDAP-1 provided a strong fluorescent signal from inside cells. They loaded human cancer cells with MDAP-1 plus 0.1, 0.2, or 0.5 mM malondialdehyde and saw that the intensity of the signal increased along with the concentration of malondialdehyde. Next, they added a solution containing 0.1 or 0.5 mM hydrogen peroxide, a reactive oxygen species that triggers oxidative stress, to cancer cells preloaded with MDAP-1. Again, the higher concentration led to a brighter fluorescent signal. As a control, they added ascorbic acid, an antioxidant that scavenges reactive oxygen species, to cells loaded with the probe and hydrogen peroxide. Those cells did not fluoresce.
Christoph Binder of the Medical University of Vienna says “this method is potentially terrific for imaging in cells but also in tissues or animals.” Indeed, Zeng says they are planning to use the probe to study malondialdehyde in prostate cancer cells and mice with prostate cancer tumors.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter