Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Origins Of Life
How the first nucleotides might have formed on Earth
Model compounds spontaneously form nucleotides under prebiotic conditions
by Celia Henry Arnaud
April 27, 2016
| A version of this story appeared in
Volume 94, Issue 18

An ongoing challenge for researchers who are studying the origins of life is figuring out how biopolymers such as DNA and RNA might have formed in the first place. In the case of RNA, a candidate for the first genetic material, researchers have observed the formation of its constituent bases and sugars under model early Earth conditions. But demonstrating how those bases and sugars might have combined to form nucleotides under prebiotic conditions has been challenging.
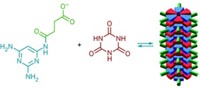
Now, a team led by Nicholas V. Hud of Georgia Institute of Technology has identified nitrogen-containing heterocycles that spontaneously react with the sugar ribose-5-phosphate in water to form nucleotides (Nat. Commun. 2016, DOI: 10.1038/ncomms11328).
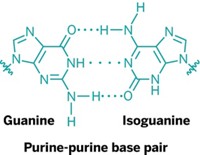
These nucleotides are capable of forming hydrogen-bonded base pairs similar to the Watson-Crick base pairs formed by modern nucleic acids. Furthermore, the newly created building blocks can self-assemble into large, stacked, noncovalent complexes, which could eventually facilitate the formation of RNA-like molecules.
Hud’s team made the nucleotides with the heterocycles barbituric acid and melamine, both of which have been observed in model prebiotic reactions in the past. Both of them reacted spontaneously with ribose with yields greater than 50%. The bases in modern nucleic acids don’t combine with ribose under the same conditions.
“We think we can consider these bases to be very plausibly prebiotic,” Hud says. “They’re doing things that look like they could get us on the road to making an RNA-like molecule.” Such a molecule could have later evolved to incorporate the bases now found in RNA, he contends.
Besides forming base pairs with each other, Hud says, the melamine and barbituric acid would also likely hydrogen bond with modern nucleobases—melamine with uracil and barbituric acid with adenine. Such “forward compatibility” would ensure that as the bases evolved, they would retain the ability to transfer genetic information.
In addition, Hud and coworkers found that the ribose in their plausible prebiotic nucleotides preferentially adopts the β-anomeric form, which refers to the conformation of the carbon in the sugar that attaches to the base. When the nucleotides stack into noncovalent complexes, the assemblies end up with twice as much of the β-anomer as the α-anomer.
The researchers don’t know exactly what’s causing the enrichment, but it happens both in the initial reaction between the heterocycle and the ribose and in the formation of the noncovalent complex. “This is very encouraging for us because the β form is the one life uses,” Hud says.
The work “demonstrates how a biologically relevant structure can spontaneously self-assemble in aqueous solution,” says David W. Deamer, an origins-of-life researcher at the University of California, Santa Cruz. “A similar process was likely to have occurred in the prebiotic environment as an important step toward the linear polymers that were necessary for life to begin.”
Figuring out how to make such linear polymers is the next step for Hud’s team. “We don’t have the liberty of using enzymes,” Hud says. “We need to get this to go with something plausibly prebiotic. It’s still a challenge, but we’re getting close.”
This article has been translated into Spanish by Divulgame.org and can be found here.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter