Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Ketamine metabolite could inspire fast-acting antidepressants
New findings indicate that hydroxynorketamine is essential for the drug’s activity in mice
by Michael Torrice
May 5, 2016
| A version of this story appeared in
Volume 94, Issue 19

Ketamine offers psychiatrists something that conventional antidepressants lack: speed. Most patients take selective serotonin reuptake inhibitors such as Prozac or Zoloft for weeks to months before they feel any improvement. But a single dose of ketamine can act within hours.
Unfortunately, the compound, used for decades as an anesthetic, comes with some downsides, including the potential for abuse as a psychedelic drug. So neuroscientists have tried to piece together ketamine’s antidepressant mechanism to help design new drugs.
A research team is now reporting that ketamine itself may not be involved in this mechanism. Studies in mice suggest instead that a hydroxylated metabolite is responsible for the antidepressant activity. This compound doesn’t block ketamine’s main target in the brain and doesn’t cause some of the drug’s known side effects (Nature 2016, DOI: 10.1038/nature17998).
“These exciting results, if confirmed by clinical studies, illuminate a clear pathway toward developing the next generation of rapid-acting antidepressants,” comments pharmacologist Irwin Lucki of the Uniformed Services University of the Health Sciences, who was not involved in the work.
In the study, scientists led by Todd D. Gould of the University of Maryland School of Medicine found that (2R,6R)-hydroxynorketamine (HNK) produced similar antidepressant effects as ketamine in mice in experiments that modeled different aspects of depression, including the loss of motivation to seek out pleasurable foods and scents after stress. Meanwhile, mice showed no signs of changes in sensory perception when given HNK, and the animals didn’t self-administer the compound, which would have been an indication of abuse potential.
To determine if the metabolite was necessary for ketamine’s antidepressant activity, the research team synthesized 6,6-dideuteroketamine, an analog that replaces two C–H bonds broken during ketamine metabolism with slightly stronger C–D bonds. “That difference is enough to make it a little harder to metabolize,” says team member Patrick J. Morris of the National Institutes of Health.
As a result, HNK levels in the brain of mice receiving the deuterated compound were lower compared with those getting ketamine. The analog produced little to no antidepressant effect in mice, demonstrating that forming HNK is necessary for ketamine’s activity.
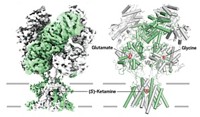
In further studies, the team determined that HNK doesn’t inhibit the NMDA receptor—ketamine’s anesthetic target in the brain. Although the researchers haven’t found HNK’s specific target, they present evidence that suggests it could act through a pathway that activates signaling from a related receptor called AMPA.
“The target of ketamine’s antidepressant activity has often been assumed to be the NMDA receptor,” Gould says. “We show that’s not the case.” He also points out that attempts to develop novel fast-acting antidepressants by inhibiting the NMDA receptor have had limited success in clinical trials.
One molecule that has shown rapid antidepressant activity and is currently in clinical trials is rapastinel. Joseph R. Moskal of Northwestern University, who is chief scientific officer at biopharmaceutical company Aptinyx, discovered rapastinel, which activates AMPA receptor signaling via NMDA receptors. He thinks the ketamine metabolite findings reinforce a shift in thinking about ketamine’s antidepressant activity from NMDA-receptor inhibition toward other, possibly AMPA-related, pathways in the brain.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter