Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Constructing an ophiobolin via radical cascade
Cytotoxic natural product succumbs to total synthesis in just nine steps
by Bethany Halford
May 30, 2016
| A version of this story appeared in
Volume 94, Issue 22
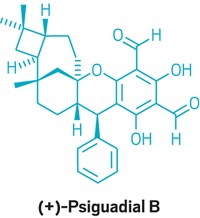
Ophiobolin sesterterpenes are a family of fungal metabolites that have been shown to have cancer-killing properties, even against the drug-resistant brain tumor glioblastoma multiforme. As targets for total synthesis, they pose plenty of challenges for organic chemists: They’re packed with stereocenters and feature a 5-8-5 fused ring system that’s tough to construct. Previous efforts have produced ophiobolin A in 47 steps and ophiobolin C in 38 steps. Chemists at the University of California, Berkeley, have now managed to make another family member, (–)-6-epi-ophiobolin N, in just nine steps, starting from farnesol (Science 2016, DOI: 10.1126/science.aaf6742). The key process in the synthetic route, developed by Thomas J. Maimone, Zachary G. Brill, and Huck K. Grover, is a radical cascade reaction that knits together the 5-8-5 fused ring system. When the researchers first tried this radical reaction, they found it gave them a mix of stereoisomers at one carbon, with the majority of the product having the undesired stereochemistry. After some trial and error, they found that addition of a complex thiol could shift the stereochemical outcome to produce more of the desired product. The researchers say this work lays the foundation for forging other complex ring systems that could benefit total syntheses.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter