Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Autonomous chemically fueled molecular motor revs up
Molecular motor runs all by itself, without external intervention, on a tank of chemical fuel
by Stu Borman
June 10, 2016
| A version of this story appeared in
Volume 94, Issue 24
Motor proteins are essential for moving muscles in animals and transporting molecules across cell membranes. Researchers have now mimicked them by designing and creating the first synthetic motor that runs autonomously—without further intervention—on chemical fuel.
Mother Nature isn’t easy to emulate, so an artificial chemically powered motor that works like a motor protein “is a major advance,” says Jonathan Clayden of the University of Bristol, an expert on chemistry-based biological mimicry. It could power motion “on a nanometer scale if attached to a polymer or solid support,” he says, and “could lead to molecular solenoids, paddles, propellers, and valves.”
Earlier nonautonomous chemically powered motors required constant intervention—they had to be fed a sequence of compounds to complete each operating cycle. Researchers have designed autonomous molecular motors, but those required a continuous source of external light instead of a tank of chemical fuel, making them more like a plugged-in electric motor than a car.
Ben L. Feringa of the University of Groningen, whose group has developed both types of earlier systems, says motors like the new autonomous chemically driven one “might ultimately have far-reaching prospects for powering nanomachines and robotics.”
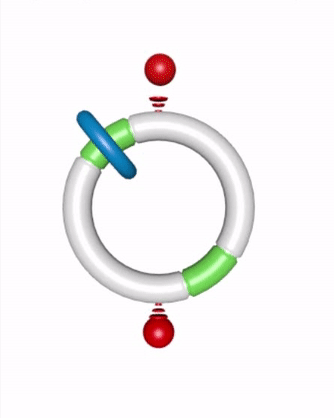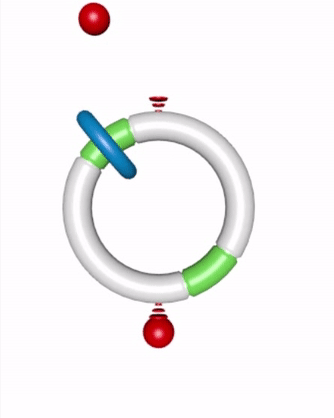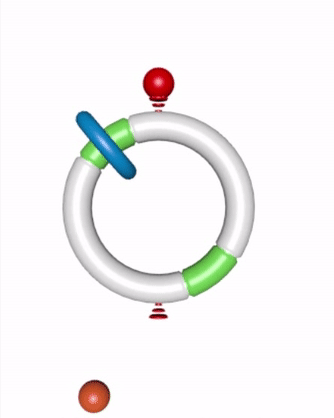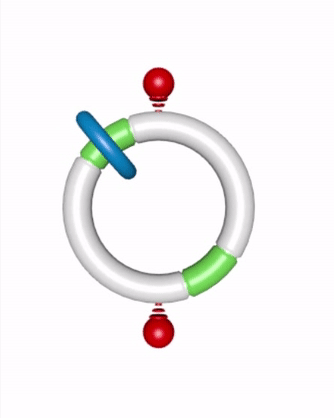
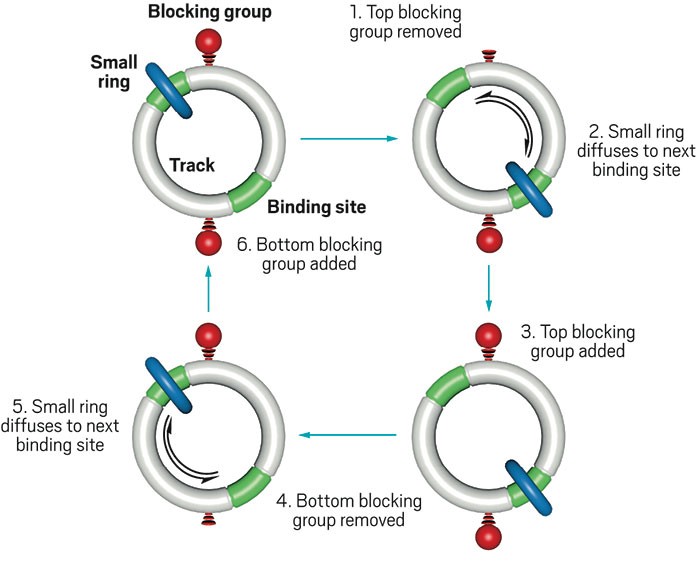
David A. Leigh and coworkers at the University of Manchester got the new motor running by synthesizing a [2]catenane, a small organic ring that is threaded onto and can travel around a track-like large ring (Nature 2016, DOI: 10.1038/nature18013). The small ring moves along the track by diffusing between two fixed fumaramide binding sites. Removable blocking groups on the track keep the small ring from moving backward, enforcing its progress as it diffuses in a forward direction.
The chemical fuel is 9-fluorenylmethoxycarbonyl chloride (Fmoc-Cl) mixed with triethylamine (Et3N). Fmoc-Cl reacts catalytically to form the Fmoc carbonate blocking groups, and Et3N cleaves them. These reactions produce dibenzofulvene, triethylamine hydrochloride, and carbon dioxide as waste materials.
The motor runs very slowly, about 12 hours per turn. “Light-powered synthetic molecular motors are much faster,” Clayden says, “so raising the rate of motion will be a key challenge to overcome.”
Leigh says that, like most first versions, the new molecular motor is hopelessly inefficient. However, “the first motor cars could only go 5 miles per hour,” Leigh says. So you have to start somewhere.”
This article has been translated into Spanish by Divulgame.org and can be found here.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter