Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Crystalline coating tames rambunctious reagents
Adamantane derivative forms inclusion compounds that make it easier to handle sensitive and smelly chemicals
by Stephen K. Ritter
September 12, 2016
| A version of this story appeared in
Volume 94, Issue 36
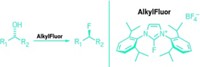
Chemistry has always been a smelly and potentially hazardous business. To safely handle useful but highly sensitive, toxic, and malodorous compounds, chemists have developed methods involving vacuum lines, glove boxes, and fume hoods. But those approaches can be costly and are often inconvenient to use. Researchers have thus come up with some clever work-arounds, such as encapsulating air- and moisture-sensitive reagents in paraffin capsules, protecting enzymes in cross-linked aggregates, and storing reactive metals in silica or alumina powders. But for organic reagents, a general-use crystalline material that acts as an inert delivery system has been missing. Alexander Schwenger, Wolfgang Frey, and Clemens Richert of the University of Stuttgart have now found one in the form of a bulkily substituted adamantane that creates inclusion compounds to tame rambunctious reagents such as benzoyl chloride, cyclohexyl isocyanide, and phosphorus trichloride (Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201605507). The researchers mix tetrakis(dimethoxyphenyl)adamantane (TDA) with the reagents to generate crystalline materials that mask the reactivity and odor of the reagents and make them easy to handle and dispense on the benchtop. When the crystal-coated materials are dissolved in an appropriate solvent, the reagents are able to react as normal, and TDA precipitates and can be recovered by filtration for reuse.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter