Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Coupling phenols and allylic alcohols
Palladium-catalyzed enantioselective oxa-Heck reaction creates chemical building blocks that are difficult to make through standard synthetic procedures
by Bethany Halford
December 5, 2016
| A version of this story appeared in
Volume 94, Issue 48
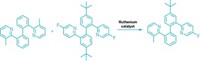
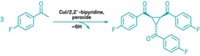
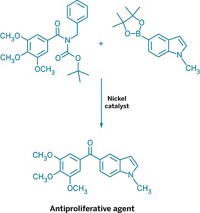
Although chemists have plenty of ways to add carbon atoms to alkenes—including the Nobel Prize-winning Heck reaction—they have fewer options when working with heteroatom nucleophiles, such as alcohols. Now, Matthew S. Sigman and his group at the University of Utah report an intermolecular oxa-Heck reaction that allows them to couple phenols with allylic alcohols in an enantioselective manner (J. Am. Chem. Soc. 2016, DOI: 10.1021/jacs.6b11486). The reaction, which is catalyzed by palladium in the presence of a chiral pyridine oxazoline ligand, creates building blocks that would be tough to make using more common synthetic reactions, such as a Michael addition (example shown). Sigman’s team selected allylic alcohols as the alkene in the reaction in order to influence which hydrogen would be removed during the β-elimination to form the chiral product. The chemists show the reaction works with a broad scope of allylic alcohols and phenols, and even successfully swaps the phenol for cumene hydroperoxide in several examples. Next, they plan to explore the scope of the reaction with other heteroatom nucleophiles that are compatible with the coupling strategy.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter