Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Chiral Lewis acid catalyzes asymmetric photocycloaddition
Reaction takes place through triplet energy transfer instead of photoinduced electron transfer
by Bethany Halford
December 19, 2016
| A version of this story appeared in
Volume 94, Issue 49
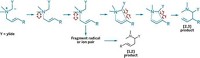
Being able to control the stereochemistry of organic reactions is important when making certain molecules, such as drug candidates or natural products. Although photochemistry allows chemists to create structural elements that are difficult to access in other ways, such as cyclobutanes, it can be tough to do photochemical reactions enantioselectively. Chemists at the University of Wisconsin, Madison, led by Tehshik P. Yoon now report an asymmetric [2 + 2] photocycloaddition of 2′-hydroxychalcones (Science 2016, DOI: 10.1126/science.aai8228). The reaction uses tris(bipyridyl) ruthenium(II) as a sensitizer and a chiral Lewis acid to catalyze the reaction (example shown). The chiral Lewis acid serves to dramatically lower the triplet energy state of the chalcone substrate. “This is an energy-transfer reaction instead of a photoredox reaction,” Yoon explains. The Wisconsin chemists expect this approach will help others apply a “detailed understanding of chiral Lewis acid catalysis to stereocontrol in reactions involving electronically excited states,” they write in their report.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter