Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Chemical Bonding
Six bonds to carbon: Confirmed
Chemists obtain elusive crystal structure of hexamethylbenzene dication, confirming its nonclassical structure with a six-coordinate carbon atom
by Stephen K. Ritter
December 19, 2016
| A version of this story appeared in
Volume 94, Issue 49
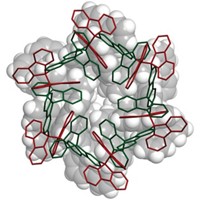
Among chemistry’s most fundamental concepts is that carbon is tetravalent and forms four bonds to other atoms. And when it comes to aromaticity, benzene’s hexagonal planar ring structure is taken as gospel. Chemists have thus been fascinated by finding exceptions to these truths. One example is hexamethylbenzene dication, C6(CH3)62+, a molecule first prepared in 1973 and suspected of not only having a six-coordinate carbon atom but also of being nonplanar. But the structure has only been inferred by spectroscopy. Moritz Malischewski and Konrad Seppelt of Free University of Berlin have now reported the first X-ray crystal structure for C6(CH3)62+, confirming it has a pentagonal pyramidal C6 framework with a six-coordinate carbon at the apex of the molecule (Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201608795). Like other nonclassical carbocations, such as CH5+ and the norbornyl cation (C7H11+), experimental proof of the C6(CH3)62+ structure proved challenging. The team first prepared an epoxide of hexamethyl Dewar benzene (a bicyclic benzene isomer) and then dissolved the epoxide in magic acid (HSO3F/SbF5) and added anhydrous HF at low temperature. Under these superacidic conditions, O2- is pulled off to generate C6(CH3)6(SbF6)2•HSO3F. Structural and computational analysis revealed that the molecule retains its aromaticity.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter