Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Catalyst flip-flops chirality to build patterned copolymer
Like a chiral switch, catalyst helps string together alternating norbornene building blocks
by Louisa Dalton
September 14, 2016

Stringing together a polymer that links individual building blocks in alternating order isn’t easy. It’s like convincing a kindergarten class to line up boy-girl-boy-girl instead of randomly holding the hand of whoever is nearest. But now, researchers have found that a catalyst offers chemists a simple way to create such polymers. The organometallic catalyst flips back and forth between two chiral conformations to build an ordered polymer of alternating, almost identical norbornenes (ACS Cent. Sci. 2016, DOI: 10.1021/acscentsci.6b00200).
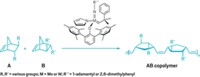
Although nature has ways to order the patterning of complex polymers, including proteins and DNA, chemists have just a handful of strategies for creating an alternating chain of two building blocks—an AB copolymer. Richard R. Schrock and coworkers at Massachusetts Institute of Technology offer a new route to such copolymers, via a ring-opening metathesis polymerization (ROMP) that relies on the inversion of a catalyst.
“Sequence control is one of the important remaining frontiers in polymer synthesis,” says Geoffrey W. Coates of Cornell University, who worked out a similar strategy in 2009 to make an AB copolymer from β-lactones (J. Am. Chem. Soc. 2009, DOI: 10.1021/ja9075327). Polymers made out of norbornenes are used in microelectronics and other high-performance polymer applications.
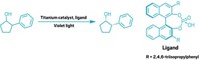
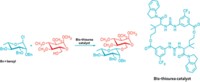
The heart of Schrock’s catalyst is a double bond connecting a four-coordinated metal atom—molybdenum or tungsten—to a carbon atom. The metal atom is also connected to a nitrogen-bearing ring and a large collection of aromatic rings that together block one side of the double bond more than the other. The catalyst exists in solution in both of its enantiomeric, or mirror image, forms. The A and B building blocks used to make the polymer are slightly different trans-2,3-disubstituted-5,6-norbornenes, and each is a pure enantiomer—chosen so that the two monomers are almost, but not exact, mirror images of each other.
When the catalyst’s metal-carbon double bond initiates ROMP, it approaches the double bond of one of the norbornenes. The catalyst naturally approaches from its least hindered side, so it first favors one monomer over the other.
As the reaction proceeds, it forms a metallacyclobutane intermediate ring, which inverts the chiral catalyst as the ring opens. In the next step, the inverted catalyst then preferentially sidles up to the other not-quite-identical monomer, and the process starts over again.
“First one way, then the other way,” Schrock says. “It is an absolutely natural part of the process.”
Schrock’s group tried about 45 reaction variations and found nine that correctly strung together AB copolymers at least 90% of the time.
“The success of this reaction depends on monomers having the same reactivity but different chirality,” Schrock says. He would love to significantly improve the catalyst’s sensitivity to the monomer’s chiral center. Then he would be able to vary the arms of the monomers more, perhaps even making one hydrophobic and another hydrophilic.
Nicole S. Sampson at Stony Brook University is intrigued by the polynorbornene backbone with alternating side chains, which is “quite distinct” from AB copolymers reported in earlier work. Controlling the structure, she says, would allow better tailoring of materials.
For any possible applications, “the kicker is that you have to use enantiomerically pure monomers,” Schrock says, which are difficult to separate. “But we’ll see. This offers a lot of potential to make polymers that have unusually precise, never-been-seen-before structures.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter