Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Potential diabetes therapy: Engineered cells that control blood sugar
Cells programmed to sense glucose and release insulin prevent hyperglycemia in diabetic mice
by Michael Torrice
December 12, 2016

People with type 1 diabetes must inject themselves with insulin multiple times per day. This is because their immune system has destroyed cells in the pancreas that secrete insulin to maintain a healthy blood glucose level.
A team of bioengineers now report a possible alternative to such injections. The researchers engineered human kidney cells to act like pancreatic β cells, namely to sense blood glucose levels and produce insulin accordingly (Science 2016, DOI: 10.1126/science.aaf4006). When implanted in mice with type 1 diabetes, the cells prevent high blood glucose levels, also known as hyperglycemia.
Right now, “all we offer diabetic patients to cope with their disease is to have them measure their blood glucose levels and then inject a hormone,” says Martin Fussenegger of the Swiss Federal Institute of Technology, Zurich, who led the team that engineered the cells. Although this works, he says, getting the dose right can be tough. “We set out to pioneer a new disease treatment concept.”
Scientists have been working on ways to restore β cells in diabetic patients. For example, doctors have tested therapies in which they transplant islet cells, which contain β cells, into patients. And other researchers have tried to derive β cells from stem cells for transplantation. But all of these potential therapies suffer from the same problem, Fussenegger says: They involve cells the immune system will eventually destroy. “β cells are not very robust cells; that’s why we have diabetes,” he says.
For that reason, Fussenegger and his colleagues decided to program a heartier cell type, the human embryonic kidney (HEK) cell, to behave like delicate β cells. To do so, they replicated the cellular circuitry that allows the pancreatic cells to respond to blood glucose levels.
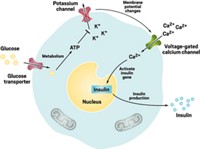
Basically, β cells regulate glucose levels with the help of three types of proteins that sit in their membranes: glucose transporters, potassium channels, and voltage-gated calcium channels. Glucose enters β cells through the glucose transporters. The cells then metabolize the glucose, producing adenosine triphosphate (ATP). As ATP levels increase, potassium channels shut down, stopping the outward flow of potassium ions from the cell. This changes the voltage across the cell membrane, which in turn activates voltage-gated calcium ion channels. Calcium ions then flow into the cell and turn on gene expression pathways that trigger insulin production.
HEK cells already have a glucose transporter and a potassium channel, so Fussenegger and his colleagues just had to engineer the cells to express the calcium channel and a calcium-dependent gene circuit for insulin production.
To test the resulting cells, the team encapsulated them in alginate beads and implanted them in mice that had their β cells destroyed chemically. The animals survived for the entire three-week experiment, while mice receiving nonengineered cells died after a few days, demonstrating that the β cell mimics could reverse insulin deficiency, which is fatal. The engineered cells also restored normal glucose levels faster than implanted human islet cells.
“Clinicians and people with diabetes will be excited by this new research and by the general notion of engineering glucose-responsive insulin-secreting cells that are not derived from pancreatic islet cells,” says John Pickup, a professor of diabetes and metabolism at King’s College London. Islet-cell-based therapies, he says, have several downsides, including high costs and side effects from the required immune-suppressing medications.
Still, Pickup says this new strategy is in its early stages. For example, the kinetics of the insulin release from the engineered cells needs further tuning to better mimic β cells. The implanted cells, he says, don’t release the hormone fast enough to keep up with the typical blood glucose fluctuations a person experiences throughout the day.
Fussenegger says his team is working on improving the function of the cellular circuit and robustness of the cells.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter