Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
New process for generating hydrogen fuel
Catalyst produces hydrogen from methanol and water at relatively low temperatures
by Stu Borman
March 22, 2017
| A version of this story appeared in
Volume 95, Issue 13

A sustainable, cleaner-burning alternative to gasoline could help shrink the carbon footprint of cars and trucks. One option is to use methanol by catalytically releasing hydrogen from the liquid to power a hydrogen fuel cell.
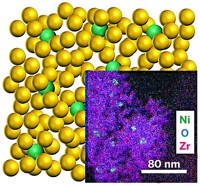
Ding Ma of Peking University, Beijing, and coworkers report a new catalyst to do so: atomically dispersed platinum over molybdenum carbide particles that drive the efficient and relatively low-temperature “reforming” of CH3OH and water to form H2 (Nature 2017, DOI: 10.1038/nature21672).
The process is about five times as efficient as the previous H2-from-methanol champ, a ruthenium-catalyzed dehydrogenation developed by Matthias Beller of the University of Rostock and coworkers (Nature 2013, DOI: 10.1038/nature11891).
The new Pt/MoC catalyst works at relatively low temperatures, 150 to 190 °C—cooler than the 200 °C or more traditionally used to reform CH3OH vapor, but considerably hotter than Beller’s process, which works at 65 to 95 °C. But the new process has advantages over the Beller technique: It avoids the use of caustic hydroxide, and the Pt/MoC catalyst is heterogeneous, which makes it cheaper and easier to recycle than the homogeneous ruthenium catalyst.
Ma calculates that a 50-L tank of CH3OH and catalyst with 6 to 10 g of Pt—about the weight of a wedding ring—could power a Toyota Mirai, a hydrogen fuel cell concept car, for about 690 km. The CH3OH would cost about $15 and the Pt about $320, but the catalyst is potentially recyclable.
Pt is relatively rare and expensive compared with other metal catalysts, but Ma points out that automobile catalytic converters now contain 1 to 4 g of recyclable noble metals, “so 8 g Pt is not a big number.”
Reaction engineer Dion Vlachos of the University of Delaware comments that the new process “has a technological edge in terms of reaction rate,” but improving long-term catalyst stability, developing means for catalyst regeneration, and finding alternatives to noble metals “are important future directions for widespread commercialization.”
Beller calls Ma’s catalyst “a major breakthrough,” noting that this type of catalyst might also be useful for other aqueous-phase reforming processes, such as those involving biowaste or ethanol.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter