Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
ACS Meeting News: Good reasons to study technetium chemistry
by Stephen Ritter
April 6, 2017

Credit: Shutterstock
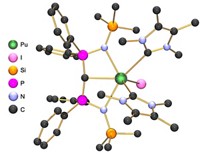
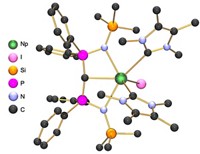
When the periodic table was being put together in the late 1800s, a hole remained in the center of the middle row of transition metals where element 43 should be. This space stayed vacant until 1937, when the invention of the cyclotron facilitated the discovery of technetium as the first and lightest artificially produced element. Being a radioactive element—it has no stable isotopes—means technetium’s chemistry has been explored less than most elements, at least until recently.
On Tuesday at the American Chemical Society national meeting in San Francisco, Alfred P. Sattelberger of Argonne National Laboratory described a decade’s worth of new research on this “hot element.” Working with Kenneth R. Czerwinski, Frederic Poineau, and their students at the University of Nevada, Las Vegas, Sattelberger has produced some 40 research papers on various aspects of technetium chemistry, including one recently that discussed how technetium’s nuclear makeup causes it to be radioactive (J. Chem. Educ. 2017, DOI: 10.1021/acs.jchemed.6b00343).
Sattelberger presented the findings during a symposium celebrating the 60th anniversary of the Division of Inorganic Chemistry. “There are three good reasons to study technetium,” he said. First, researchers need to know how Tc compares with its next-door neighbors regarding its synthetic and structural chemistry.
Related story: Making technetium-99m
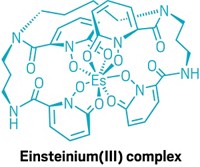
Second, the radioisotope Tc-99m, with a half-life of six hours, is a workhorse in diagnostic medicine for imaging the heart and other organs. Sattelberger described how researchers study compounds made with Tc-99, which is the decay product of Tc-99m and has a half-life of 212,000 years, before trying to make analogs with the short-lived Tc-99m.
The third reason is that about 6% of the fission products of U-235 nuclear fuel rods end up as Tc-99, Sattelberger noted. Studying Tc-99 compounds is helping sort out the best options for long-term storage of spent radioactive fuel.
Sattelberger and his colleagues have shown that, with radioactive compounds, simple experiments become difficult and a tour de force approach is needed to study them, commented Debbie C. Crans of Colorado State University, one of the symposium’s organizers. This team has shown, however, that “it is possible for inorganic and nuclear chemists to unlock the box of fundamental technetium chemistry so we can begin exploring what else can be done with these compounds.”
Other related stories:


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter