Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Business
India promotes industry-academia biopharmaceutical R&D
by K. V. Venkatasubramanian, special to C&EN
June 12, 2017
| A version of this story appeared in
Volume 95, Issue 24
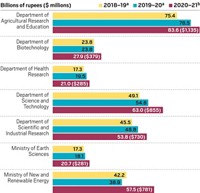
India has approved the first-ever industry-academia partnership program to accelerate discovery research and enable development of products such as vaccines, biotherapeutics, and diagnostics. Called the Biopharma Mission, it is aimed at improving the health of India’s population through affordable product development, a goal in line with Prime Minister Narendra Modi’s government’s recently released National Health Policy. The new effort, which includes training a skilled workforce, is expected to enhance India’s technological and product development capabilities in the biopharmaceutical sector so it becomes globally competitive in the next 10–15 years, the government says in a statement. The program will aim to enhance technology transfer capabilities in the public and private sectors and enable start-ups and small and medium-size enterprises to build the capacity for sharing innovative research. The $250 million initiative, which includes $125 million in a loan from the World Bank, is expected to roll out by October. The Biotechnology Industry Research Assistance Council, a government enterprise under the Department of Biotechnology, will implement it. Renu Swarup, senior adviser at the department, says the mission “will facilitate translation of research leads from research institutes to product development.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter