Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Policy
Tensions build as U.S. chemical law turns one year old
EPA scrambles to implement changes required under the updated Toxic Substances Control Act
by Britt E. Erickson
June 26, 2017
| A version of this story appeared in
Volume 95, Issue 26

A year ago, U.S. chemical manufacturers and environmental advocates were celebrating the enactment of amendments to the Toxic Substances Control Act—the law that governs new and existing chemicals in the U.S. market. They had high expectations that the changes would help boost consumer confidence in the safety of chemicals sold in the U.S.
But one year later, the chemical industry and environmental advocates are at odds over how the Environmental Protection Agency should implement the revised law. Tensions are high over a number of issues, including EPA’s review of new chemicals and how the agency should handle confidential business information. The two sides also disagree on whether EPA should use its new authorities to regulate certain uses of the solvent trichloroethylene and two paint-stripping chemicals, N-methyl-2-pyrrolidone and methylene chloride.
New chemicals delayed to market
One of the unintended consequences of the TSCA overhaul was a temporary backlog of new chemicals waiting to be evaluated by EPA. Before the law was modernized, the agency typically had about 300 new chemicals under review at any given time. That number peaked at about 600 in January, according to EPA.
The backlog arose because the new law requires EPA, for the first time, to affirm the safety of each new chemical before the substance enters the market. EPA must determine whether the substance presents, may present, or is not likely to present an unreasonable risk, or whether there is insufficient information to conduct a reasonable evaluation.
EPA initially interpreted the law to mean that it should evaluate all “reasonably foreseeable uses” of a chemical when making its risk determinations. Chemical manufacturers said that approach was delaying new chemicals from entering the U.S. marketplace and stifling innovation.
EPA has since changed its process to include only uses of a new chemical intended by the original manufacturer. If the agency believes that the chemical could be used in ways other than the manufacturer intended, it will regulate those applications simultaneously through what are called significant new use rules, says Jeffery Morris. He directs the agency’s Office of Pollution Prevention & Toxics, which manages EPA’s programs under TSCA.
The new process will eliminate the backlog by the end of July, EPA predicts. The agency now has about 430 new chemicals under review and is putting extra staff on the job temporarily until the number drops to about 300, which EPA considers its baseline for substances under review, Morris says.
The chemical industry supports EPA’s latest approach, but manufacturers say it won’t solve all the delays they face getting new chemicals to market. Since the new law was enacted last year, EPA has issued more requests for toxicity data and more restrictions than it did under the old TSCA, several companies note.
Makers of specialty chemicals are raising concerns about the recent spike in the number of new chemicals denied exemptions. EPA used to generously grant exemptions to substances produced in amounts of less than 10,000 kg/year. Substances that are approved for these low-volume exemptions undergo a 30-day review instead of the typical 90-day evaluation for new chemicals. Makers of substances given low-volume exemption do not have to pay user fees to the agency for the review.
In general, the process of getting new chemicals onto the market is taking longer than it did before the amended law was enacted, says Dan Newton, senior manager of government relations at the Society of Chemical Manufacturers & Affiliates. This trade group represents the specialty chemical industry.
EPA is now denying more requests for low-volume exemptions and regulating more new chemicals than under the old TSCA, Newton says. “This is going to impact the introduction of greener and safer chemicals,” he claims. That’s because companies are increasingly introducing chemicals that have been designed to be safer and better for the environment than the substances they are intended to replace.
Environmental advocates, meanwhile, argue that chemical manufacturers should expect that more of their new products will be subject to restrictions on commercialization or will require additional toxicity tests. “The new law requires EPA to issue such orders whenever it either lacks sufficient information to evaluate a new chemical, or makes a risk- or exposure-based finding that indicates potential concern,” says Richard Denison, a lead senior scientist with the environmental group Environmental Defense Fund. “In such cases, the orders must impose conditions sufficient to mitigate the concern,” he explains in a recent blog post.
EPA issued consent orders restricting commercialization of about half of the new chemical submissions that it has processed since the updated TSCA took effect, Denison estimates. That number was 8% before the changes occurred, according to EPA.
“While both EPA and the regulated community have suffered growing pains in implementing these immediately effective requirements of the law, the picture is far less bleak than industry representatives continuously claim in public,” Denison says. “Many of their assertions and demands are simply not accurate or not consistent with or allowed by the new law.”
EPA’s Morris is encouraging chemical manufacturers to have what he calls “robust conversations” with the agency before they submit their new chemical applications. Such engagement, coupled with a guidance document that EPA is developing for companies, should go a long way toward getting future assessments of new chemicals completed within 90 days, he says.
Year 1 of the new chemical law, by the numbers
EPA regulations are on the rise for new chemicals.
▸ 401 new chemical reviews completed
▸ 87 substances, or 22%, allowed to be commercialized without restrictions, down from ~87% before June 22, 2016
▸ 195 chemicals, or 49%, allowed to be commercialized with restrictions, up from 8% before June 22, 2016
▸ 3, or 1%, not allowed to be commercialized until manufacturer provides additional toxicity data, up from 0 before June 22, 2016
▸ 116, or 29%, withdrawn by manufacturer, up from 5% before June 22, 2016
Note: Percentages do not total 100% because of rounding.
Source: EPA
Trade secrets versus public’s right to know
As EPA scrambles to get its new chemicals program back on track, the agency is trying to resolve another thorny issue related to a provision under the revised TSCA involving confidential business information (CBI). The provision is intended to balance the need to protect disclosure of CBI with the public’s right to know what chemicals are in consumer products.
Under the revamped law, EPA is required to assign a “unique identifier to each specific chemical identity” that the agency approves as CBI. The agency must also “apply that identifier consistently to all information relevant to the applicable chemical substance,” including nonconfidential information.
EPA is wrestling with how to apply the same unique identifier to both CBI and related publicly available information reported under TSCA in a way that ensures that trade secrets, including the chemical identity of the substance, won’t be disclosed.
During a public meeting in late May, EPA officials highlighted scenarios in which applying the same unique identifier to all information pertaining to a substance could cause a chemical’s identity to be revealed. The agency presented two potential ways to avoid inadvertent disclosure of a chemical’s identity that is approved as CBI and encouraged companies and the public to offer other suggestions.
In EPA’s first approach, all nonconfidential information about a chemical substance would be kept confidential. EPA acknowledges that such an approach goes against the intent of the revised TSCA, which aims “to limit CBI protection to a relatively narrow set of circumstances.” As a result, EPA hasn’t gotten much enthusiasm for its first option.
Under the second approach, EPA would assign a unique identifier for each submission related to a chemical, but only for submissions made by the same manufacturer. If a different company submits information about the substance, the chemical would be assigned a different identifier. The public would be able to connect some, but not all, information about the substance.
In general, manufacturers support the agency’s second approach. This option is the only one from EPA that achieves the requirements of the law, says Christina Franz, senior director of regulatory and technical affairs at the American Chemistry Council, an industry group representing chemical manufacturers.
Environmental groups, however, are raising concerns about EPA’s interpretation that the law’s provision for a unique identifier creates a problem. “EPA finds an apparent conflict that does not exist in the law, and it overstates a problem that it has successfully navigated in the past,” EDF’s Denison said at the meeting.
He and other environmental advocates warned EPA not to hinder the public’s ability to know what chemicals they are being exposed to. “If a company wants to mask its association with the chemical, that association can be protected through masking the company’s identity rather than the chemical identity,” Denison suggested.
EPA is accepting comments from the public on its two alternatives and other potential approaches for assigning unique identifiers until July 7.
High-risk uses of three solvents face uncertainty
One of the big drivers of TSCA reform last year was chemicals on the market for which little to no toxicity data exist. While Congress negotiated legislation to update the chemical control law, EPA evaluated the risks of a handful of selected chemicals using its authority under the old TSCA.
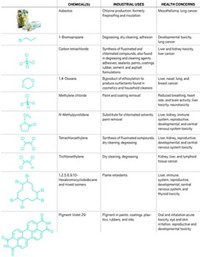
EPA flagged about 90 substances that it considered high priority for review and possible regulation because of their high potentials for hazard and exposure. In the final days of the Obama Administration, EPA proposed rules to restrict certain high-risk uses of three of the chemicals. One is trichloroethylene (TCE) in aerosol spray degreasers, spot-cleaning agents in dry cleaning, and vapor degreasing. The other two are methylene chloride and N-methyl-2-pyrrolidone (NMP) in paint and coating removers.
Those three compounds also appear on EPA’s list of 10 chemicals facing evaluation under the revised TSCA. By law, EPA must complete its evaluation of these 10 chemicals by November 2019.
Chemical manufacturers are urging EPA to scrap the Obama-era assessments, which concluded that specific uses of the three chemicals pose risks to human health, including cancer and neurodevelopmental effects. They are pushing EPA to instead review the three chemicals as part of the agency’s upcoming assessment of the first 10 substances under the amended TSCA.
Environmental groups argue that EPA should finalize the proposed restrictions as soon as possible to protect U.S. workers and consumers.
“Congress expressly authorized EPA to take early actions under the amended TSCA based on broad agreement that such actions were necessary to demonstrate the new law was working and to begin to restore confidence in the federal chemical safety system,” says Jennifer McPartland, a senior scientist in EDF’s health program. “We strongly urge the agency to finalize its proposed bans on high-risk uses of TCE, methylene chloride, and NMP.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter