Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Straightening out natural products’ stereochemistry
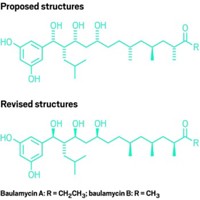
Multipronged method used to correct the structures of baulamycins A and B
by Bethany Halford
July 26, 2017
| A version of this story appeared in
Volume 95, Issue 31
When chemists at the University of Bristol set out to make a pair of natural products with antimicrobial activity, it seemed like a quick and easy way to highlight some synthetic methodology their lab had developed. But after finishing their 10-step syntheses of baulamycins A and B, the nuclear magnetic resonance (NMR) spectra of the molecules they made didn’t match those originally reported for the natural material. It turns out that the stereochemistry of the original compounds had been incorrectly assigned, and the Bristol team had been making the wrong molecules all along .
The team, led by Varinder K. Aggarwal, Eddie L. Myers, and Craig P. Butts, now report a method to figure out the correct stereochemistry of baulamycins A and B, and other complex molecules.
It’s not so easy, Aggarwal says. Baulamycins A and B have an open chain structure with seven stereocenters. “It was very daunting to think about which of the seven stereocenters was incorrect,” Aggarwal notes. There are, after all, 128 possible isomers.
The researchers used computational methods and NMR spectra of the natural material to determine the most likely configuration of the three contiguous stereocenters on one side of the molecule. They then made a mixture of four possible isomers of baulamycin A in predetermined unequal ratios, making it possible to quickly identify which compound’s spectra matched that of natural baulamycin A. The chemists then synthesized the correct structure on its own (Nature 2017, DOI: 10.1038/nature23265).
“The method we’ve described could be very useful for the structural determination of other complex molecules,” Aggarwal says.
Rajib K. Goswami, a professor at the Indian Association for the Cultivation of Science who also synthesized the incorrect structures of baulamycins A and B, says the multipronged approach used by the Bristol team is an important contribution. He adds that the work highlights how total synthesis of natural products allows chemists to confirm the accuracy of proposed structures, something he says is “vital for any industry interested in developing new natural-product-based drugs.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter