Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Water
What’s that Stuff
What are pool chemicals, and how do they protect swimmers?
Chlorine kills microbes in pools, but don’t blame it for your green hair
by Celia Henry Arnaud
August 1, 2017
| A version of this story appeared in
Volume 95, Issue 32

People love to complain about chlorine in pools—often as much as they enjoy plunging into the clean water. Chlorine-based compounds can dry out skin, turn eyes red, and produce the familiar and pungent pool smell.
On the other hand, thanks to chlorine, we can swim in water free of microbes, such as Escherichia coli, that can cause digestive troubles. In addition to disinfectants such as chlorine, pool operators also add chemicals to control the pH, alkalinity, and hardness of the water.
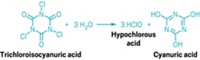
The chlorine used to disinfect pools is rarely present as elemental chlorine (Cl2). It’s typically added as part of more complex molecules such as hydrogen- and oxygen-containing hypochlorites or carbon-, nitrogen-, and oxygen-containing isocyanurates, explains Thomas M. Lachocki, chief executive officer of the National Swimming Pool Foundation. When these compounds are added to water, they spontaneously form hypochlorous acid, which is the disinfecting agent commonly called free chlorine in pool lingo.
Residential pool owners are most likely to use a form of chlorine best known as trichlor or trichloro-S-triazinetrione because it dissolves slowly, has high chlorine content, and is easy to use, Lachocki says. Getting the concentration of the disinfectant right is a balancing act. The concentration needs to be high enough that some disinfectant is always in the water. But it also needs to be low enough to be comfortable for swimmers. Adding a stabilizer such as cyanuric acid helps protect the hypochlorous acid from degrading in sunlight.
Chlorinated compounds aren’t the only options for first-line disinfectants. Bromine-containing compounds that generate hypobromous acid can also be used to kill pathogens. But they’re used in hot tubs more often than in swimming pools because bromine is more stable than chlorine at the high temperatures used in hot tubs.
Newer pool hygiene systems take extra steps to kill chlorine-resistant microbes that can cause intestinal infections. Some recreational facilities treat for Cryptosporidium, which has a protective coat that makes the microbe hard to destroy with chlorine, says Blake Stark, general manager of water treatment chemicals and filtration media at NSF International, a standard-setting and certification organization.
For such purposes, ultraviolet radiation in the filtration system has become popular. Although it’s particularly good for inactivating Cryptosporidium, UV doesn’t make a good stand-alone disinfectant, Lachocki says. “It doesn’t maintain a residual in the water and only inactivates and oxidizes in the UV chamber,” he says. That means that microbes brought into the water by swimmers aren’t effectively killed if UV is the only method used.
But UV water treatment has another benefit: It can prevent that potent “chlorine” smell associated with indoor pools. Swimmers are partly to blame for the stink. That odor is created when chlorine reacts with compounds in urine and sweat to form trichloramine. UV light can degrade these compounds. In addition to causing pool smell, trichloramine and other disinfection by-products may be linked to asthma in swimmers.
Pool disinfectants work optimally when the water pH is between 7.2 and 7.8. So pool operators must add another class of compounds to maintain this pH range and push the equilibrium between hypochlorous acid and hypochlorite ions toward the acid, which is a better disinfectant, Lachocki says. Typical chemicals used include muriatic acid (an archaic name for hydrochloric acid), sodium bisulfate, carbon dioxide, sulfuric acid, and sodium carbonate. Sodium bicarbonate also works, but it has a greater effect on the total alkalinity of the water than it does on the pH.
And all these pool chemicals are big business. According to Pkdata, a market research firm that collects data on the swimming pool and spa industry, there are more than 8.5 million residential pools—in ground and aboveground—in the U.S., not including Alaska and Hawaii. More than 719 billion L of water get treated to fill those pools. As a result, the market for the chemicals used in that treatment is roughly $2 billion.
Pool chemistry certainly produces a lot of greenbacks, and sometimes even a green pool. When the diving pool at the 2016 Summer Olympics in Rio de Janeiro turned a shade of emerald, it was possibly because of algae growth or an excess of a copper-containing algicide. The actual culprit will never be known conclusively because Olympic officials drained and refilled the pool without running chemical tests.
On occasion, pool chemistry is also to blame for a bit of green hair. Urban myths would have you believe that chlorine causes unwelcome green highlights. But the change of hue is thanks to copper-containing algicides added to water or copper found in the water used to fill the pool. Sometimes the copper even originates from corroded plumbing. It’s like the green patina that appears on copper-plated buildings, except it’s on your head.
Thankfully, if folks follow directions on pool chemical labels—which are regulated in the U.S. by the Environmental Protection Agency—these debacles can usually be avoided. Or it may be time to get your water tested for copper.
Update: This article was modified on Aug. 1 to refresh its information and data.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter