Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
A new molecular star is born
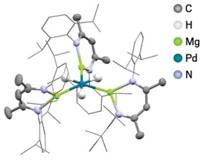
First reported pentanuclear heteroleptic palladium coordination complex takes on the shape of a truncated star
by Stephen K. Ritter
August 7, 2017
| A version of this story appeared in
Volume 95, Issue 32
One thing chemists have learned over the years is that geometry is innate in molecular structures. Besides regular polygons, unexpected shapes often emerge when researchers explore new combinations of metal and ligand components. In one of the latest examples, a team led by Dillip Kumar Chand of Indian Institute of Technology Madras has built a “truncated molecular star,” a pentagram-shaped palladium coordination complex (Chem.–Eur. J. 2017, DOI: 10.1002/chem.201702264). Most self-assembled coordination complexes are built from a metal coordinated with one type of ligand (homoleptic). Complexes made using two or more types of ligand (heteroleptic) are not as common. On top of that, the ligands often are chelating, meaning the ligand binds one metal through two or more atoms. Despite the success in geometrical self-assembling coordination chemistry, Chand and coworkers discovered that their new molecular star is unique not just because of its pointless shape—a pentagon ringed by isosceles trapezoids—but also because it appears to be the first heteroleptic, nonchelating, multicompartment supramolecular coordination complex built with five palladium atoms. The researchers assembled the compound from a palladium(II) salt paired with equal amounts of 1,4-phenylenebis(methylene) diisonicotinate and 4,4′-bipyridine ligands in a one-pot reaction at room temperature. The chemistry behind the cool-looking truncated star molecule isn’t “pointless,” the researchers say, as their study provides new insight on how intricate coordination complex structures form and could lead to guest-binding applications that mimic multicompartmental biological systems.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter