Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
At last, cancer-fighting inhibitors hit deubiquitinase target selectively
Two research groups find small molecules that block the enzyme USP7 and show promise in mice with tumors
by Stu Borman
October 19, 2017
| A version of this story appeared in
Volume 95, Issue 42
In the body, ubiquitinating enzymes regulate proteins—marking them for disposal, for example—by adding ubiquitin peptides to them. And deubiquitinases (DUBs) refine regulation by removing ubiquitins. But DUBs can also promote tumor growth and curb anticancer immune responses. Researchers have been trying to find small molecules that fight cancer by inhibiting DUB activity, but progress has been slow.

In two new studies, researchers have passed a milestone in the field by identifying some of the most promising inhibitors yet for USP7 (ubiquitin-specific protease-7), the most common anticancer target among the approximately 100 types of DUBs found in the body. USP7 can promote cancer by reducing levels of the tumor suppressor protein p53 and by impairing the immune system’s ability to detect and eliminate tumors. Inhibiting USP7 can have the opposite effect, killing cancer cells.
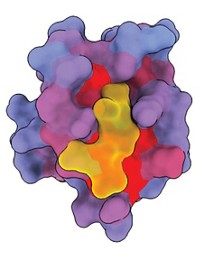
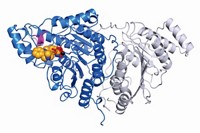
However, previous inhibitors blocked USP7 essentially by only a single mechanism: bonding covalently or binding noncovalently to the enzyme’s active-site cysteine. Scientists haven’t obtained, or at least haven’t published, small-molecule/USP7 crystal structures to guide drug discovery. And selective USP7 binding, with its potential for reduced side effects, has been elusive because many DUBs have similar structures and active sites.

In the new studies, two research groups found the most selective USP7 inhibitors yet reported, and enzyme crystal structures they obtained reveal that the compounds bind at novel sites. Till Maurer and Ingrid E. Wertz of Genentech and coworkers used nuclear magnetic resonance spectroscopy-based screening and structure-based design to identify and optimize GNE-6640 and GNE-6776, which inhibit USP7 selectively compared with 36 other DUBs (Nature 2017, DOI: 10.1038/nature24006). The compounds block USP7 far from the active-site cysteine the enzyme has in common with other DUBs and kill tumors in cell culture and in live mice.
Stephanos Ioannidis of Forma Therapeutics, Andrew P. Turnbull of Cancer Research UK Therapeutic Discovery Laboratories, David Komander of the MRC Laboratory of Molecular Biology, Benedikt M. Kessler of the University of Oxford, Sylvie Urbé and Michael J. Clague of the University of Liverpool, and coworkers screened a Forma compound library to identify and refine the noncovalent inhibitor FT671 and the covalent inhibitor FT827 (Nature 2017, DOI: 10.1038/nature24451). The compounds block USP7 but not 38 other DUBs. They target USP7 at or near its active site but also interact with a uniquely structured pocket, and they inhibit the proliferation of cultured cancer cells and the growth of tumors in mice.
The groups used structural biology “to fine-tune inhibition of USP7 in a way that exploited differences between USP7 and other DUBs, maximizing the chances of obtaining high selectivity,” comments Michael Mattern, vice president of corporate affairs at Progenra, which is developing USP7 inhibitors. “How these findings translate to clinical utility, however, remains to be determined.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter