Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Discovery
Looking beyond opioids for safer pain relief
Researchers explore many avenues to treat chronic pain but don’t see a single, clear path forward
by Bethany Halford
November 13, 2017
| A version of this story appeared in
Volume 95, Issue 45
COVER STORY
Looking beyond opioids for safer pain relief
Pain has been a part of Kate Mitchell’s life since she was 10 years old. What began as an ache in her ankle spread throughout her body until doctors eventually diagnosed her with a collection of disorders that cause chronic pain, including rheumatoid arthritis and fibromyalgia. Now in her 20s, Mitchell does her best to juggle life as a graduate student in literature with a schedule packed with medical appointments—sometimes as many as eight per week—and the routine that helps her keep chronic pain at bay. Her daily regimen to manage her illnesses and treat the accompanying pain consists of doing yoga, walking, and taking 40 pills, including two doses of acetaminophen mixed with the opioid hydrocodone.
On her best days, she lives with a moderate level of pain she describes as dull and achy. On her worst, the pain is like a hot poker pressed against her joints.
That’s when she reaches for her emergency pain medication: oxycodone combined with acetaminophen. The pill brings physical relief but some mental discomfort. The ongoing opioid crisis has created a stigma around chronic use of prescription pain relievers. Mitchell says sometimes people think she’s taking her medication just to get high.
“What I think a lot of people don’t understand is that people with chronic pain don’t take medication only when it gets to the worst pain possible,” Mitchell says. “I take the pain medication so it doesn’t get to a worst-case scenario.”


Mitchell is uncomfortable with the stigma associated with taking opioids and wouldn’t mind switching to a different class of pain reliever. But she’s wary. “I’m terrified of it not doing anything for my pain,” she says.
Yet opioids provide relief to only a fraction of people who, like Mitchell, live with chronic pain, according to doctors and experts. And long-term use can have dangerous consequences, ranging from addiction to accidental overdose. Researchers are looking to move beyond opioids for safer, nonaddictive, and more effective treatments for pain. In their search, scientists are exploring a wide range of drug targets beyond the mu opioid receptors that oxycodone and other opioids bind to and trigger. These alternative targets include G protein-coupled receptors, ion channels, enzymes, and growth factors.
While many of these avenues look promising, experts in the field caution that obstacles stand in the way of making a good treatment for chronic pain.
“All chronic pain is not the same. We’re assuming it is,” says Kathleen Sluka, a pain expert at the University of Iowa. Although chronic pain—commonly defined as pain lasting longer than three months—can begin with an injury or be caused by a disease like fibromyalgia or arthritis, people with chronic pain tend to get lumped together in terms of diagnosis and treatment. “I think that’s been our biggest mistake in pain research,” Sluka says.
Relief, at a cost
As many as 100 million adults in the U.S. have chronic pain, according to a 2011 report from the National Academies’ Institute of Medicine. Most of these people have no effective way to relieve their pain.
“The toolbox for treating chronic pain is really fairly limited,” says Gary Brenner, a professor of anesthesia at Harvard Medical School who sees patients at Massachusetts General Hospital’s Pain Management Center. “Every day I work in the clinic, I take care of many terribly suffering individuals because we really don’t have an adequate armamentarium of therapeutic products. You meet people whose lives have been destroyed, really completely destroyed, by a pain problem.”

Although opioids are known to be good for treating short-term, acute pain, there is little in the medical literature to suggest that opioids are effective for chronic pain. What’s more, the medication may actually be detrimental. A 2014 report from the Agency for Healthcare Research & Quality concluded that “evidence on long-term opioid therapy for chronic pain is very limited but suggests an increased risk of serious harms that appears to be dose-dependent.”
“We certainly need to keep working from the chemistry and basic research angles to try to find better alternatives to opioids,” says Dave Thomas, who works in the Pain Research & Analgesia Development Program of the National Institute on Drug Abuse (NIDA).
“What happens with opioids, especially in the context of chronic pain, is that patients take these drugs for considerable periods of time,” says Ajay Yekkirala, cofounder and chief scientific officer of Blue Therapeutics, a company developing pain relievers.
People taking opioids for extended periods rapidly build up tolerance to them and have to take higher doses to get the same pain relief, he says. That’s when opioids become dangerous. In addition to their analgesic effect, high doses of opioids also cause respiratory depression, in which a person’s breathing slows down or stops.
“We cannot hope to prevent opioid misuse and overdose without addressing the treatment needs of people with moderate-to-severe chronic pain,” wrote Nora D. Volkow, director of NIDA, and Francis S. Collins, director of the National Institutes of Health, in the New England Journal of Medicine in July. “Though more cautious opioid prescribing is an important first step, there is a clear need for safer, more effective treatments.”
Dimming the pain
Some of the most advanced efforts to develop safer treatments have been focused on designing better molecules to target G protein-coupled receptors (GPCRs). This large family includes opioid receptors and cannabinoid receptors. With a better understanding of how those receptors work, chemists now think they can design molecules to parse the analgesic effect from dangerous side effects.
GPCRs are embedded within cell membranes, where they transmit messages regulating a number of biological functions, including inflammation and neurotransmission.
Their central role in processing so many functions makes them both attractive and tricky targets for scientists developing treatments for pain. “GPCRs are not simply ‘off’ or ‘on,’ ” says Kenneth Jacobson, a medicinal chemist who studies GPCRs at the National Institute of Diabetes & Digestive & Kidney Diseases. They’re more like dimmer switches, with different conformations of the receptors turning on different pathways. “If you can make an agonist that will stabilize one conformation and not another, you can activate a single signaling pathway and not activate the undesired pathway,” he says.
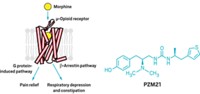
One advanced strategy being pursued by drug developers is to design molecules that more selectively modulate known pain receptors—namely, the mu opioid receptor targeted by morphine and fentanyl.
The aim of such compounds is to turn on the G protein pathway, which causes pain relief, while avoiding the β-arrestin pathway, which is linked to constipation and respiratory depression. One advanced example of a drug candidate that uses this approach is Trevena’s Olinvo (oliceridine), which the U.S. Food & Drug Administration is currently reviewing as a treatment for acute pain. But whether this strategy will work for chronic pain has yet to be proved.
Scientists are also looking to selectively target the kappa opioid receptor, which is associated with pain and can cause undesirable side effects in the central nervous system, such as hallucinations. Cara Therapeutics’ compound CR845 was designed to activate this receptor only in the peripheral nervous system, outside the brain. That’s because it isn’t very good at crossing the blood-brain barrier. The compound is currently in Phase IIb clinical trials to treat chronic pain associated with osteoarthritis.
But opioid receptors aren’t the only GPCRs associated with pain. Jacobson, for example, works on A3 adenosine receptors, which, when activated, can reduce pain signals en route to the brain. In collaboration with Saint Louis University’s Daniela Salvemini, his lab is working to create compounds that can turn on this receptor and in doing so reduce and prevent neuropathic pain, a type of chronic pain caused by damage to nerve fibers. People being treated for diabetes or taking chemotherapy for cancer can also develop neuropathic pain.

The cannabinoid receptors, known as CB1 and CB2, are also promising GPCR targets for treating pain. Scientists know that the cannabinoid tetrahydrocannabinol (THC), the active ingredient in cannabis, can partially activate these receptors, producing analgesic effects.
But long-term THC use can also slow one’s thinking and impair memory. “There are a lot of efforts to develop cannabinoids without side effects,” says Alexandros Makriyannis, a medicinal chemist and director of the Center for Drug Discovery at Northeastern University. Makriyannis estimates that his lab has produced about 14,000 compounds that target cannabinoid receptors for various uses, including analgesia.
“Pain comes in different flavors,” Makriyannis says. “There are types of pain that can most effectively be dealt with through the CB1 receptor and other types through the CB2 receptor. Each receptor has a different way of approaching pain, and there are some cases where both of them are involved.” One example of a drug candidate that turns on the CB2 receptor to relieve pain is Arena Pharmaceuticals’ APD371, currently in Phase IIa clinical trials for treating pain.
Beyond GPCRs
If GPCRs are dimmer switches that can dial back pain, then ion channels control electrical circuits that underlie these neuronal signals. Researchers think that by locking these channels into a state where they can no longer open or close, it might be possible to stop a pain signal headed for the brain.

Genetic studies have shown that the voltage-gated sodium channel Nav1.7 is overactive in people who have certain rare pain disorders and is inactive in people who can’t feel pain at all. This finding has made the channel, and related channels Nav1.8 and Nav1.9, attractive to drugmakers. Convergence Pharmaceuticals (which was acquired by Biogen) developed raxatrigine. The drug is in Phase II clinical trials for chronic pain caused by a disease affecting a nerve that transmits pain to the face. Pfizer also had a compound that targeted this receptor in Phase II clinical trials, but it didn’t prove to be effective and was discontinued.
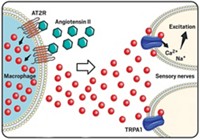
Christa Müller, a medicinal chemist at the University of Bonn, has been exploring compounds that halt the ligand-gated ion channel P2X4 as potential analgesics. Adenosine triphosphate (ATP) activates these channels, inducing and sustaining inflammatory pain.
The challenge to making effective compounds for ion channels, she says, is striking the right balance between potency, selectivity, solubility, and the ability for a compound to cross into the central nervous system.
“The binding site of ATP in P2X4 is quite polar,” Müller points out. It contains a lot of positively charged amino acids to bind to ATP’s triphosphate group. Consequently, she says, it’s difficult to block that site. Instead, Müller’s group is working on compounds that bind to another spot on P2X4 but still prevent ATP from activating the ion channel.
The most widely used painkillers, aspirin and ibuprofen, don’t target GPCRs or ion channels. Instead, these nonsteroidal anti-inflammatory drugs, or NSAIDs, go after a family of enzymes called cyclooxygenases (COXs). In theory, scientists could eliminate side effects associated with GPCRs and ion channels, which exist throughout the body, by inhibiting an enzyme that was produced only after an injury and leads to inflammation or pain. In practice, however, the underlying biochemistry can be more complicated: Many COX-2 inhibitors were pulled from the market in 2004 after they were found to increase the risk of heart attack and stroke.
Drugmakers have also been pursuing inhibitors of nerve growth factor as nonopioid painkillers. Nerve growth factor levels, which are higher in a number of chronic pain conditions, are known to mediate pain via interaction with tropomyosin receptor kinase A. Scientists have hypothesized that blocking this interaction could provide pain relief.
Advertisement
Antibodies that sequester nerve growth factor have been the most promising potential therapies in this area, but they’ve also run into problems. In 2010, FDA halted clinical trials of these antibodies because of reports they were causing joints to deteriorate and osteoarthritis to rapidly progress.
FDA eventually removed the restriction on testing these therapies, and tanezumab, an antibody from Pfizer and Eli Lilly & Co., is currently in Phase III clinical trials to treat chronic low back pain and chronic pain from osteoarthritis.
Even with new pain therapies on the horizon, experts caution that a single treatment won’t work for all types of chronic pain.
“When I teach students about pain mechanisms, one of the key messages is that you are wired to make sure that you can detect pain,” says Donna Hammond, a professor of anesthesia and pharmacology at University of Iowa’s Carver College of Medicine. “The system has so much redundancy in it that if you block one pathway, there is something else that can help make sure the message comes through.”
Hammond notes that many of the efforts to develop new pain treatments focus on a single channel or a single receptor. “Perhaps instead of that specificity, we need to be looking at drugs with a broader spectrum of activity,” she says.
COVER STORY
Looking beyond opioids for safer pain relief
“We probably are not going to get a magic bullet treatment,” NIDA’s Thomas agrees. “What we will hopefully get is a number of different drugs that may work for some people.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter