Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Breaking isopropyl’s symmetry with C–H activation
Palladium-ligand combo locks up isopropyl moiety to generate chiral molecules from feedstock derivative
by Bethany Halford
February 2, 2017
| A version of this story appeared in
Volume 95, Issue 6

When it comes to classic human beauty, people seem to prefer symmetry in facial features and figures. But chemists synthesizing drug candidates doggedly pursue asymmetry in their molecules. That’s because asymmetric molecules, or chiral compounds, have distinct properties arising from their stereochemistry. Being able to make such stereogenic centers directly saves chemists time and resources.
Chemists at Scripps Research Institute California and Bristol-Myers Squibb now report a way to break the symmetry of an isopropyl group using C–H activation chemistry (Science 2017, DOI: 10.1126/science.aal5175). Previously, such a feat of desymmetrization has been accomplished only by enzymes, notes Scripps’s Jin-Quan Yu, who spearheaded the project.
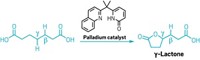
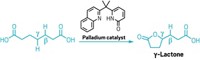
Yu’s group uses an amide derivative of the feedstock chemical isobutyric acid as a substrate. When combined with palladium and a chiral bidentate ligand, one methyl group of the isobutyric acid’s isopropyl moiety selectively undergoes C–H activation and then reacts with an aryl, vinyl, or alkynyl iodide. This process forms an α-chiral center in the amide derivative.
The trick, Yu says, was to design a ligand that could lock the substrate’s methyl groups into just the right position to react the way the chemists wanted them to. “You can’t let them rotate,” Yu says. “You can’t let them slip away from the desired conformation.”
“This work by Jin-Quan Yu and coworkers solves one of the toughest problems of catalytic enantioselective C–H activation: the intermolecular desymmetrization of methyl groups,” comments Olivier Baudoin, a transition metal catalysis expert at the University of Basel. “Yu’s work takes us to levels of simplicity and efficiency that up to now have been the prerogative of natural systems.”
“Yu and his team have accomplished a major advance in the enantioselective construction of chiral, α-substituted carboxylic acid derivatives,” adds Scott Denmark, an expert in stereocontrol in C–C bond-forming reactions at the University of Illinois, Urbana-Champaign. “By harnessing the C–H bond functionalization of sp3 carbons with aryl, alkenyl, and alkynyl residues, these investigators have opened a fundamentally new avenue to accessing these highly privileged motifs.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter