Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Modifying methionine on proteins
Redox chemistry turns reticent amino acid into bioconjugation handle
by Bethany Halford
February 9, 2017
| A version of this story appeared in
Volume 95, Issue 7

Thanks to the chemistry of its thiol group, cysteine has long been the go-to amino acid when researchers want to modify a protein. It’s easy to pluck off cysteine’s thiol proton and replace it with something else, such as a drug molecule or a dye.
But this chemistry can cause problems, explains Christopher J. Chang, a chemical biologist at the University of California, Berkeley. Those cysteine residues are often involved in catalysis or play a role in stabilizing the protein via formation of disulfide bonds. If scientists could find a different amino acid to modify, they’d be less likely to disrupt protein function when making antibody-drug conjugates or preparing covalent protein probes.
Teaming up with synthetic chemists in F. Dean Toste’s lab at UC Berkeley, Chang’s lab has managed to do just that: develop a bioconjugation method that selectively modifies methionine residues (Science 2017, DOI: 10.1126/science.aal3316).
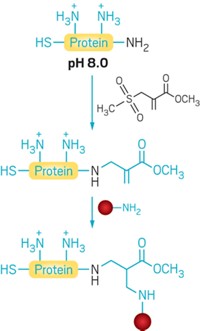
The method is based on the residue’s redox chemistry. Under physiological conditions, the chemists found that methionine reacts with oxaziridine reagents to form a sulfimide (shown). From there, the researchers can create more elaborate structures using click chemistry, for example.
In bioconjugate chemistry, “You’re trying to modify something but not perturb the function,” Chang says. Because methionine is a thioether and relatively chemically inert compared with cysteine’s thiol, modifying methionine is less likely to change the function of a protein.
“Adding methionine to the arsenal of amino acids that can be targeted for selective chemical tagging opens the door to a variety of applications in antibody bioconjugation and functional proteomics,” comments Eranthie Weerapana, an expert in biochemical probes at Boston College.
Toste says the work shows how it’s possible to use organic chemistry to solve a biological problem. “This is a nice example of what happens when synthetic chemists talk to chemical biologists.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter