Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Surprising amyloid structure solved
Bacterial peptide forms amyloids with α-helices instead of β-sheets; new structures could be targets for antibiotics
by Celia Henry Arnaud
February 23, 2017
| A version of this story appeared in
Volume 95, Issue 9

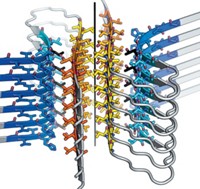
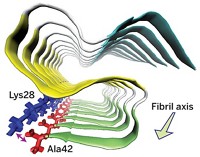
When Meytal Landau, a structural biologist at Technion—Israel Institute of Technology, recently solved the structure of some peptide fibrils she believed were amyloids, she nearly fell out of her chair. She expected to see the β-sheet architecture that’s common to all known amyloid fibrils. Instead, she saw the first known α-helical version (Science 2017, DOI: 10.1126/science.aaf4901).
Amyloid fibrils are protein aggregates that pack together to form long strands. Many of these fibrils are involved in diseases, such as the amyloid-β plaques in Alzheimer’s and the α-synuclein aggregates in Parkinson’s. The β-sheet structural motif had been so ubiquitous among amyloids that researchers thought it was required in order for the aggregates to form. But that may not be the case.
PSMα3 is the most cytotoxic member of a family of peptides secreted by the bacterium Staphylococcus aureus. It’s an example of a “functional amyloid,” one that is beneficial rather than harmful to the organism that produces it. Involved in disintegrating human cells, PSMα3 is a major contributor to S. aureus’s virulence.
Landau and coworkers expected PSMα3 to be an amyloid because it formed elongated, unbranched fibrils that bind the amyloid-indicator dye Thioflavin T. Yet they wrestled to make sense of their X-ray crystallography data for months. “At first when I figured out that the structure is helical, I was mad that I had spent all this time,” Landau says. She thought she had solved the wrong form of the peptide—a monomer rather than a fibril. Only later did she realize she was looking at an α-helical version of an amyloid fibril.
During their studies, the researchers learned that PSMα3 is most toxic to cells in its fibrillar form. So Landau plans to use the newly acquired structure to design inhibitors of the amyloid as potential antibiotics. Inhibiting formation of the PSMα3 amyloid could provide a way to decrease virulence without threatening the bacterium’s life. Thus, the bacterium would be less likely to develop antibiotic resistance.
The study makes clear that amyloid-like fibrillar structures can be made from different secondary structures, says Wilfredo Colón, a chemist at Rensselaer Polytechnic Institute who studies protein aggregation. “One should not be surprised if a new amyloid-like structure is discovered containing both α and β structures or with the ability to form both types of fibril.”
“I never saw this coming,” says Daniel Otzen, a biophysicist at Aarhus University who studies protein fibrillation. “We can perhaps expect more surprises like this as we continue to delve into functional amyloids. They are truly evolutionarily optimized structures, not just pathetic failures like pathological aggregates, and nature will use whatever works.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter