Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Making macrocyclic compounds for DNA-encoded libraries
Adding magnesium enables ring-closing metathesis reactions on DNA-tagged molecules
by XiaoZhi Lim
July 6, 2017

A simple tweak to a tool for making macrocyclic compounds could help increase the diversity of DNA-encoded libraries used by drug developers to rapidly screen and identify promising drug candidates (Bioconjugate Chem. 2017, DOI: 10.1021/acs.bioconjchem.7b00292).
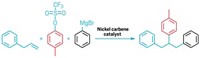
Building such libraries involves attaching short, unique DNA sequences to small molecules and then reacting those DNA-tagged building blocks together to create myriad products, which are tagged with additional unique DNA sequences. The chain of DNA markers serves as a sort of bar code to identify the compounds in a library that successfully bind to a particular drug target and to trace their synthesis history. However, these libraries generally have not been able to include ring compounds in the drug screening because transition-metal catalysts essential for ring-closing reactions are incompatible with DNA. These catalysts are central to many important organic transformations such as olefin metathesis, but they can bind to charged DNA backbones and cause the strands to fall apart.
A team led by Xiaojie Lu and Lijun Fan at GlaxoSmithKline has found that by protecting the DNA tags with magnesium ions, they can produce a variety of DNA-encoded heterocycles and macrocycles using ruthenium-catalyzed ring-closing metathesis. The team hypothesizes that because the magnesium ions occupy all the DNA’s binding sites, the ruthenium catalyst is forced to react with the substrates instead of the DNA. Preliminary tests to perform cross-metathesis reactions between two DNA-tagged primary alkenes to produce a secondary alkene were also successful.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter