Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Custom catalyst quantifies effect of nanoconfinement
Findings may be used to improve performance of nanoporous catalysts by tailoring size and shape of hollow channels
by Mitch Jacoby
January 29, 2018
| A version of this story appeared in
Volume 96, Issue 5

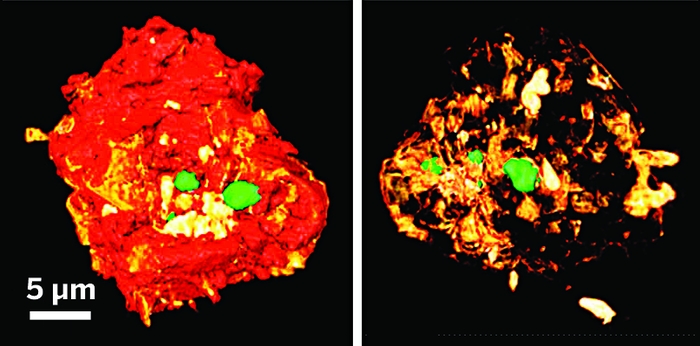
Catalytic materials with nanosized pores and channels play a central role in industrial chemistry. For example, zeolites, a family of nanoporous aluminosilicates, are widely used in petrochemical refining to produce fuels and chemicals. Researchers have long known that confining reagent molecules within nanosized channels of zeolites and other porous materials tends to enhance catalytic reactions. But the complexity of these materials makes it difficult to separate mass transport phenomena from reaction kinetics, which limits basic understanding of these systems. So Wenyu Huang of Iowa State University and Ning Fang of Georgia State University and coworkers designed model catalysts to bypass those difficulties. The materials consist of a solid silica core (100 nm in diameter) decorated with 5-nm-diameter platinum particles; the core is encapsulated within a porous silica shell (120 nm or 80 nm in diameter). The team used those catalysts and a fluorescence microscopy technique that provides single-molecule resolution to track thousands of molecular diffusion and reaction events individually. In the test reaction, a nonfluorescent molecule oxidizes on the platinum particles and forms a highly fluorescent product. By comparing the porous catalysts with nonporous reference catalysts, the team found that nanoconfinement slightly reduces rates of molecular transport, adsorption, and desorption but boosts reaction rates by about a factor of seven, a finding that may aid design of more effective nanoporous catalysts (Nat. Catal. 2018, DOI: 10.1038/s41929-017-0021-1).



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter