Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Business
How encapsulation is taking root in the laundry room
Microcapsules are increasingly being used to get fragrance and other molecules onto fabrics
by Michael McCoy
January 29, 2018
| A version of this story appeared in
Volume 96, Issue 5

A walk down the supermarket’s or big-box store’s laundry products aisle reveals several liquid detergents touting “bleach alternative.” One of them, Tide Plus Bleach Alternative HE Liquid, was in 2017 deemed the best U.S. laundry detergent at getting out stains (but second overall) by Wirecutter, a product review website owned by the New York Times Co.
But what does bleach alternative mean? Procter & Gamble, the maker of Tide, doesn’t exactly say, though it does point to enzymes that whiten and brighten clothes. The detergent also includes an optical brightener that makes clothes appear brighter by absorbing ultraviolet light and reemitting it in the blue region.
What is clear is that Tide Plus Bleach Alternative HE Liquid does not contain bleach. P&G and other detergent makers tend not to use actual bleaches because they are temperamental and reactive.
Consumer bleach comes in two main varieties, and both are problematic. Halogen-based bleaches like sodium hypochlorite can’t be incorporated in liquid detergents without prompting unwanted reactions with other ingredients. Oxygen-based bleaches can be used in powdered detergents, but they must be jump-started with temperature or activators. And in the U.S., at least, powders have fallen out of favor with consumers.
Wouldn’t it be great to be able to add an effective bleaching agent to a liquid laundry detergent? Kept on idle in the bottle, it would rev up once it mixes with the wash water and go to work on hard-to-remove stains like ink or blood.
That’s a promise of encapsulation, the practice of enrobing ingredients in a protective shell to be released when—and only when—needed. Applied to halogen- or oxygen-based oxidizing agents, the result would be a cleaning product that combines modern surfactants and enzymes with the old-fashioned power of bleach.
It’s a dream of cleaning product formulation chemists, but one that has yet to be realized. Undaunted, some companies are advancing new encapsulation-based methods for getting bleach and other cleaning ingredients to coexist.
Others see encapsulated bleach as a pipe dream. Instead, they’re content to focus on expanding the tried-and-true use of encapsulation in cleaning: keeping fragrance molecules from going down the drain during the wash cycle so they stay on clothing and keep them smelling fresh.
One of the believers in encapsulated bleach is Battelle Memorial Institute, the big Ohio-based nonprofit. Battelle scientists work mostly for corporate and government clients, but they can apply for money to work on side projects with commercial potential.
For chemist Ram Lalgudi, that project was controlled release of a compound in paint that mitigates corrosion before it can spread. Lalgudi soon realized that his controlled-release polymers could also encapsulate other active ingredients, making them available on demand through a change in conditions such as pH, temperature, or chemical environment.
Jon Cartlidge, Battelle’s business development manager for consumer and industrial solutions, is now trying to bring Lalgudi’s technology to applications such as cleaning. “Tell us an active and the trigger, and we can design a capsule to meet that need,” he says.
The technology uses polymers such as polyurethane, polymethyl methacrylate, and polylactic acid, a biodegradable resin. And although fragrances, enzymes, and biocides can all be encapsulated for the cleaning products industry, Battelle focuses on bleach.
Hydrogen peroxide is the active ingredient in liquid nonchlorine bleaching products like Clorox 2. Sodium percarbonate, which releases hydrogen peroxide when dissolved, does the job in the powdered version of Clorox 2.
The catch is that hydrogen peroxide isn’t an effective bleach at temperatures below 60 °C. Clorox 2 powder contains an activator, tetraacetylethylenediamine, that reacts with hydrogen peroxide to form peracetic acid, a more effective bleach. But the activator can’t be added to a liquid bleach without triggering a premature reaction.
How microencapsulation works
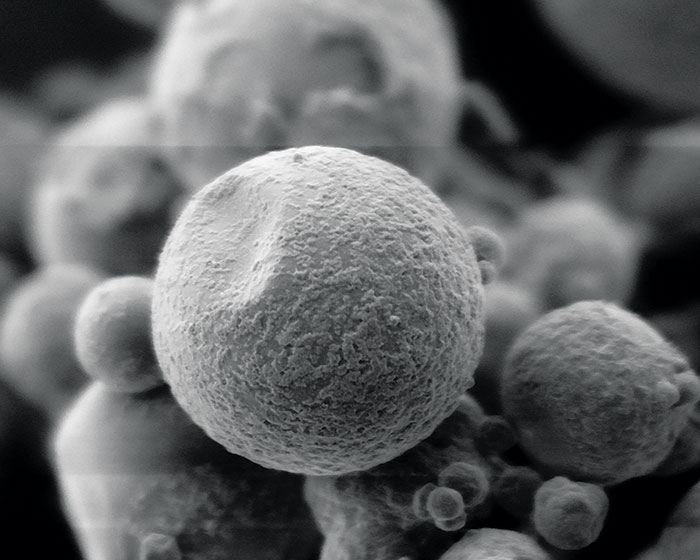
Most sources point to National Cash Register’s 1954 launch of carbonless paper as the first commercial use of microencapsulation. The encapsulated ink in the paper allowed stores and offices to do away with cash register ink ribbons and messy carbon paper.
The NCR process, called coacervation, is still used today. In it, the ingredient to be encapsulated is dispersed in a polymer-rich aqueous solution. The solvent characteristics of the medium are then changed, causing the polymer to undergo a phase separation. The polymer-rich phase spontaneously deposits itself as a coating around the dispersed ingredient and is solidified into a microcapsule, typically about 10 μm in size.
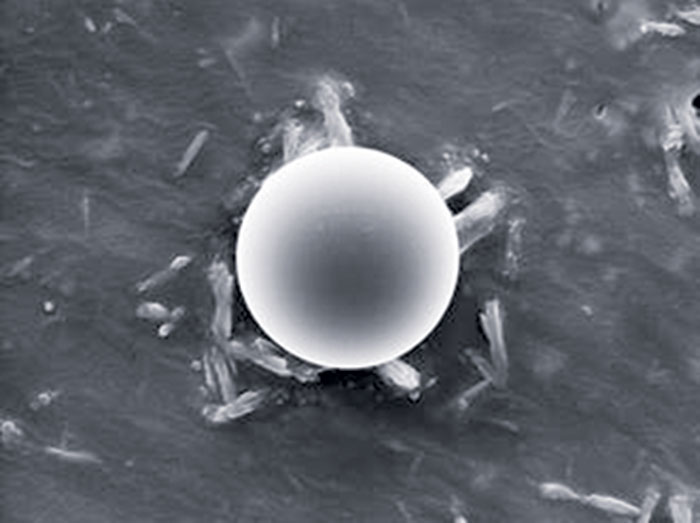
In interfacial polymerization, a monomer is dissolved in the ingredient to be encapsulated, and this solution is dispersed in an aqueous phase. A reactant to the monomer is added to the aqueous phase, causing polymerization to ensue at the surface of the ingredient and forming capsule walls. Capsules are typically 20–30 μm.
A third common technique is in situ polymerization. It’s similar to interfacial polymerization, except that no reactants are added to the ingredient. Typically, an emulsion is created of water and an oily ingredient to be encapsulated. A water-soluble resin is added and dispersed. The pH is then reduced, initiating polycondensation and yielding resins that deposit at the oil-water interface. Capsules are 5–20 μm.
Sources: Microencapsulation Innovations, Microtek Laboratories
That’s where encapsulation comes in. Battelle can encapsulate the activator, Cartlidge says, allowing it to be added to a hydrogen peroxide-based bleach. The encapsulating polymer is designed to open upon dilution in the wash water, releasing the activator to initiate the desired reaction.
Cartlidge says the approach has attracted interest but no commercial applications yet.
Itaconix, a U.K. polymer chemistry start-up, is taking a different approach to bleach. Rather than encapsulate the activator, it is encapsulating the bleach itself using what it calls vinyl alcohol-based polymers.
Since last year, the big chemical maker Solvay has been marketing a version of the bleaching agent 6-phthalimido-peroxyhexanoic acid (PAP) that is encapsulated using Itaconix technology, according to Itaconix CEO Kevin Matthews. PAP is an effective low-temperature bleach that doesn’t need an activator, Matthews notes, but it does benefit from encapsulation for stability.
PAP tends to be used in powdered industrial and institutional cleaning products. The next challenge, Matthews says, is liquid detergents for the consumer market.
In 2015, Itaconix licensed to Solvay its technology for encapsulating sodium percarbonate for highly concentrated liquid laundry and automatic dishwasher detergents. With the percarbonate thus protected, the bleach activator can be added on its own in nonencapsulated form.
The technology is still under development, Matthews says, but the potential is considerable. “Consumers say that liquid products tend to underperform relative to powders because there’s no bleach in there,” he says.
Battelle and Itaconix are relative newcomers to the encapsulation business and have chosen bleach as a high-profile target to make their marks. Established names are playing it more conservatively, staying focused on improving the delivery of fragrances and other easier-to-encapsulate molecules.
Those markets have borne fruit in the past and promise more growth in the years ahead, according to Eduard Poels, a chemist who heads the fragrance delivery center of excellence at the big Swiss flavors and fragrances firm Givaudan.
Getting clothing to smell fresh after laundering is no easy task. Fragrance molecules are typically oils, the very things that detergents are designed to remove in the wash. After washing comes drying, often by heating, and fragrance molecules tend to evaporate in the process.
Fabric softeners, added to the wash after the detergent, are an easier entry point for fragrance. Seeking more fragrance firepower, pretty much every fabric softener maker has introduced encapsulated fragrance over the past decade, Poels says.
Protected by microcapsules, the fragrance stays on fabrics through the cleaning process and releases its scent over time. The German household products giant Henkel, for example, now touts fabric softeners that keep “laundry fresh and fragrant for up to 10 weeks.”
Encapsulation has also penetrated the powdered laundry detergent market, Poels says, where polymer capsules keep fragrance oils from evaporating while in the box. Some are triggered to open in the wash water; others stay closed and are burst by friction—during folding, ironing, or wearing—after the wash.
Liquid laundry detergents are more of a technological challenge because capsules must survive what Poels calls “the extractive nature” of such products. Moreover, fragrance capsules don’t stay suspended in dense liquid detergents as easily as they do in lower-density fabric softeners.
Still, Givaudan says it has succeeded in developing encapsulation systems for highly concentrated liquids, including laundry pods. “We have products for all of them,” Poels says.
Henkel, which declined to speak to C&EN for this article, boasts of using encapsulation to enhance its Purex with Crystals Fragrance liquid detergent. “New fragrance encapsulation technology keeps clothes fresh for 30 days,” the firm’s website trumpets.
Givaudan won’t say if it’s the source of Henkel’s capsules. But Jeroen Lindhout, head of Givaudan’s global delivery systems business, calls his firm the industry leader in encapsulation for fabric care. “Over the past 10 years, the number of facilities we operate and the partners we work with have more than doubled,” he says.
Today, Givaudan operates several centers around the world where it creates fragrance microcapsules for customers. Its latest one is set to open later this year in Singapore. Poels is based at an encapsulation research center in Argenteuil, France.
For the fabric care market, melamine- formaldehyde polymers are Givaudan’s mainstay encapsulation chemistry. “Melamine-formaldehyde is the most forgiving,” Poels says. “It’s the easiest to make a performing capsule.”
Formaldehyde has become a bad word at big U.S. retailers such as Walmart and Target; it’s one of eight chemicals that Walmart has asked its suppliers to remove from their products. Poels counters that free formaldehyde from encapsulation resins is present in finished products in part-per-billion quantities, less than in many common fruits and vegetables. “There’s no risk to the health of the consumer,” he says.
Still, Givaudan executives are aware that perception can drive formulation decisions, so Poels’s team has developed formaldehyde-free encapsulants based on materials such as hydrocolloids and polyurea resins. All the firm’s encapsulation systems for the personal care industry are formaldehyde-free, Poels says, and it is beginning to offer alternatives to home care customers as well.
Seeing a trend, the specialty chemical maker Ashland has assembled an entire family of formaldehyde-free encapsulants. Most appropriate for the fabric care industry, according to Ben Sales, business manager for Ashland’s encapsulation group, is Captivates A, a line of acrylate polymers that the firm launched last year on the basis of research conducted at its acrylate innovation center in Bradford, England.
The Bradford team developed the line with the goal of offering encapsulants free of formaldehyde or isocyanates, used to make polyurea. “But after all our sensory evaluation tests,” Sales says, “we realized we haven’t just succeeded in removing those ingredients; we’ve actually succeeded in creating a technology that’s superior to what’s available.”
Melamine-formaldehyde chemistry still dominates encapsulation for the home care market, Sales acknowledges, but the tide may be turning. Ashland’s new encapsulants are already commercial in a fragrance-boosting laundry additive sold in North America, he says.
Competing against specialty chemical makers such as Ashland and fragrance makers like Givaudan are a handful of encapsulation specialists, the largest of which is Encapsys. Based in Appleton, Wis., the firm has a lineage that goes back to 1954, when National Cash Register worked with a company then known as Appleton Coated Paper to introduce encapsulated ink to the world in the form of carbonless cash register paper.
The successor to Appleton Coated went bankrupt last year, a victim of digitalization, but not before selling its Encapsys microencapsulation business to a private investment firm for $208 million in 2015.
At the time of the sale, Encapsys had annual sales of $61 million. Since then, according to Executive Director Mike Friese, the company has moved into a new headquarters building that includes laboratories and a pilot facility. As a stand-alone firm it has enjoyed continued sales growth, he says.
In 2008 Encapsys became the first to encapsulate fragrance for fabric softeners on a large scale, through an agreement with P&G. At the time, urea- and melamine-formaldehyde resins were the main encapsulation materials. Encapsys has since broadened its palette, Friese says, and can now encapsulate with polyurea and acrylate chemistry. It also uses natural products such as gelatin.
Advertisement
Carbonless paper is in its sunset years, but J. C. DeBraal, Encapsys’s director of technology, sees opportunity for encapsulation in many other markets, home and fabric care among them. In addition to fragrance, antimicrobial molecules, phase-change materials, odor absorbers, and softeners get encapsulated. Encapsys has successfully encapsulated bleach and bleach activators, DeBraal says, though the technology isn’t commercial.
In all cases, Friese adds, the key to success is working with customers to assess whether encapsulation makes economic sense. But when encapsulation helps expensive ingredients get where they need to go, the economics often do work out.
“There’s a balance that you have to hit between technology, compatibility, economics, and consumer benefit,” Friese says. “We’re adjusting all those levers. Oftentimes we find that just the fact that we’re encapsulating more than pays for itself.”





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter