Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Watching heme in live organisms
Microscopy method captures heme dynamics in Caenorhabditis elegans worms
by Celia Henry Arnaud
February 19, 2018
| A version of this story appeared in
Volume 96, Issue 8

Although heme, the oxygen-carrying organometallic compound found in red blood cells, is involved in many biological processes, little is known about how this cofactor moves around. This lack of knowledge is primarily due to the difficulty in watching heme trafficking in live organisms.
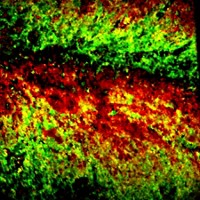
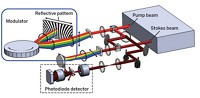
Ji-Xin Cheng of Boston University, Iqbal Hamza of the University of Maryland, and coworkers watch heme dynamics in real time using a method called transient absorption (TA) microscopy (Anal. Chem. 2018, DOI: 10.1021/acs.analchem.7b05046). This method uses a pair of laser beams at different wavelengths to measure excited-state dynamics of a chromophore. The spectroscopic signatures produced by TA allow researchers to distinguish different states of a molecule.
“Heme is an excellent molecule for TA imaging,” Cheng says. Although heme absorbs strongly, it fluoresces weakly, which means conventional fluorescence microscopy methods don’t work well with the molecule.
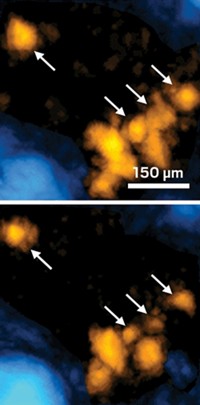
The researchers used TA microscopy to map heme dynamics in Caenorhabditis elegansworms that were fed a heme-containing compound. The images revealed that C. elegans stores heme as granules in membrane-enclosed structures called lysosomal-related organelles. The researchers used TA microscopy to watch large granules break down into smaller granules. The method is sensitive enough to detect heme at 9-µM concentrations.
Wei Min, a biological imaging expert at Columbia University, calls the work a major advance in the ability to noninvasively observe heme in living systems. Even more exciting, he says, is the potential to differentiate various states of heme, such as oxidized or reduced heme.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter