Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Latest artificial glucose-binding receptor is best yet
Supramolecular structure binds glucose strongly and selectively, pointing to future incorporation into diabetes treatment devices
by Celia Henry Arnaud
November 14, 2018
| A version of this story appeared in
Volume 96, Issue 46
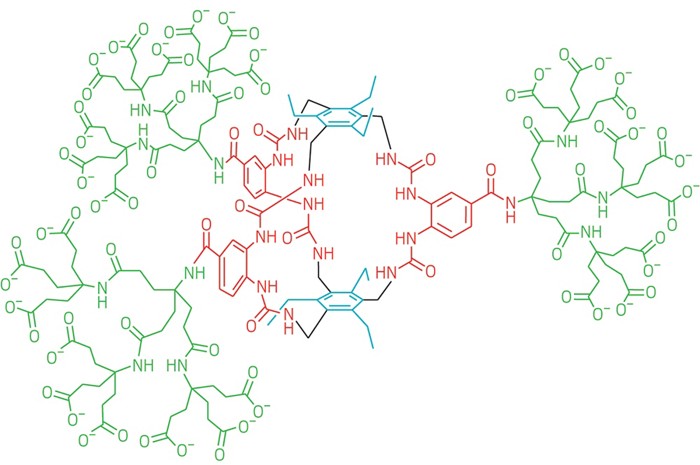
Researchers have been trying for years to make an artificial receptor to which glucose binds strongly and with high selectivity. Such a receptor molecule could be used in glucose-monitoring devices or insulin-dispensing devices that respond to glucose levels in the body. But scientists have had only modest success with their syntheses.
Now, a team at the University of Bristol and the start-up Ziylo report a synthetic receptor that binds glucose with better affinity and selectivity than previous molecules used for binding carbohydrates (Nat. Chem. 2018, DOI: 10.1038/s41557-018-0155-z). The new receptor binds glucose more than 100 times as strongly as it binds closely related sugars such as mannose or galactose.
Advertisement
When asked what differentiates his team’s new receptor from previous ones reported by his group and others, team leader Anthony P. Davis of the University of Bristol says, “It works.”
Eric V. Anslyn, a supramolecular chemist at the University of Texas, Austin, says “the work is a tour-de-force in molecular design of synthetic receptors,” adding that the “selectivity for glucose is astounding.”
Davis and his team rationally designed the receptor to contain a hollow core with six urea groups as “walls” connecting a pair of triethylmesitylene groups as a “floor” and “ceiling.” These aromatic caps are arranged to form CH-π interactions with glucose when it’s captured in between. The core is surrounded by three dendrimers that make the whole receptor soluble in water. Graduate student Robert A. Tromans synthesized the receptor.
In previous designs, the aromatic floor and ceiling were separated with isophthalamide spacers rather than urea groups. Davis played around with the design on the computer and came up with the urea spacers, which widen the core and make room for more hydrogen-bonding interactions between the receptor and glucose. The urea groups are positioned such that each one forms two hydrogen bonds with oxygen atoms on the glucose molecule.The design “went straight from computer to practice,” Davis says.
“From a supramolecular perspective, the evolution of receptor designs culminating in the current receptor is a textbook example of insightful, iterative molecular design,” says Bradley D. Smith, a supramolecular chemist at the University of Notre Dame. “The structural innovation to increase the distance between the aromatic roof and floor elements of the receptor structure, which allows an increased number of hydrogen bonding units in the sidewalls, is ingenious.”
The new receptor “is certainly the best synthetic receptor developed so far for glucose, in terms of binding constant and selectivity,” says Evgeny Kataev, a supramolecular chemist at the Technical University of Chemnitz.
The Bristol team assigned the patent for the receptor to Ziylo, a spinout company. In August, Novo Nordisk, a Danish pharmaceutical company with an emphasis on diabetes, bought Ziylo to get the patent, which it plans to use in its efforts to make glucose-responsive insulin molecules.
But Davis’s team isn’t done with the molecule yet. The researchers have started another company called Carbometrics that’s licensing the patent back from Novo Nordisk to develop the receptor for glucose monitoring. Technology of that sort already exists, but “we think we can improve it by incorporating our molecule,” Davis says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter