Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Reconstructing hemoglobin’s ancestors to trace development of structural complexity
Cooperative oxygen binding could have arisen from formation of tetramer
by Celia Henry Arnaud
May 21, 2020
| A version of this story appeared in
Volume 98, Issue 20

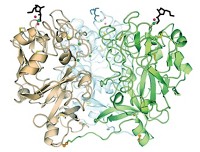
How proteins evolved the ability to form complexes made up of multiple subunits—and what functions this new structure enabled in early multimeric proteins—isn’t well understood. To better understand how such changes might have happened, Joseph W. Thornton of the University of Chicago and coworkers used ancestral protein reconstruction to trace the evolutionary pathway of the oxygen-transporting protein hemoglobin (Nature 2020, DOI: 10.1038/s41586-020-2292-y).
Modern hemoglobin forms a heterotetramer with two α subunits and two β subunits. The researchers used computational methods to infer the ancestral amino acid sequences of each of those subunits, as well as the more ancient precursor from which both α and β likely originated. They engineered bacteria to produce the proteins and characterized them with mass spectrometry and size-exclusion chromatography. When expressed together, the α and β subunits formed hemoglobin-like tetramers, but their ancient precursor formed only dimers.
By introducing mutations that happened during hemoglobin’s history into the reconstructed ancestral protein sequences, and having the bacteria produce them, the researchers found that just two changes at an interface between the subunits were sufficient to cause tetramers to form. They also found that the formation of tetramers causes cooperative binding, in which the binding of oxygen increases the hemoglobin’s affinity for additional oxygen molecules.
The researchers suggest that the ease of evolving structural complexity and cooperativity may not be limited to hemoglobin. The team plans to use this method to study whether other proteins also evolved multimeric structures and cooperative binding with relatively few mutations.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter