Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biochemistry
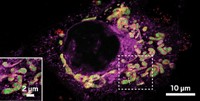
New labeling method can reveal cell-cell interactions
A bacterial enzyme, transformed via directed evolution, offers a way to tag cells that come into close contact with one another
by Esther Landhuis
February 12, 2019

Tracking interactions between different types of cells in the body can offer clues to an organism’s development, reveal cellular functions, or track tissues’ responses to treatments. However, existing methods involve toxic reagents or require engineering proteins on each of the interacting cells, which means scientists have to know beforehand which cells are involved. Now, researchers have devised a scheme to label cell-cell encounters in their natural environment without prior knowledge of which type of cells interact (J. Am. Chem. Soc. 2019, DOI: 10.1021/jacs.8b10286).
Peng. R. Chen of Peking University was studying a bacterial enzyme called sortase A when he realized it had the potential to be a useful tool for monitoring such interactions. In the natural world, sortase A fortifies bacteria by cross-linking proteins in their cell walls. The enzyme does this in a specific manner: it finds a signature amino-acid sequence in one protein and connects it to a signature sequence of three to five glycines in another protein. In the lab, researchers have taken advantage of the enzyme’s ability to recognize these specific sites in order to modify proteins in a variety of ways, both in vitro and in living cells.
In a study published last January, a team led by Rockefeller University immunologist Gabriel D. Victora developed a sortase-based approach to track immune cell interactions. First, they engineered mice that express sortase A on the surface of T cells and the multi-glycine sequence in the membrane proteins of B cells or dendritic cells. Then they injected the mice with a peptide tagged with biotin, which gets captured by sortase A. When a T cell comes close enough to a B cell or dendritic cell for the enzyme and multi-glycine site to interact, the biotin tag gets transferred to the interacting cell (Nature 2018, DOI: 10.1038/nature25442). By tracking such cell-cell interactions, this system is able to reveal the initiation of immune responses in vivo.
The sortase A-producing cells can only reveal interactions with cells that were pre-engineered to produce a three- to five-glycine tag on the cell surface. Chen wanted a tool to reveal as-yet-unknown interactions between cells. His solution: make sortase A more promiscuous.
Using a Nobel Prize-winning technique that applies natural selection to reshape enzymes, Chen’s team evolved a sortase A variant that can bind to proteins containing just a single glycine at the N-terminus rather than needing a sequence of three to five glycines. N-terminal glycine is found in membrane proteins of virtually all cells. With this broader scope, Chen says, “you can label any cell of interest.”
Advertisement
So far his team has used the system, called EXCELL (enzyme-mediated proximity cell labeling), to track interactions among kidney, cancer, and B cell lines. Next, they will express the enzyme in immune cells or tumor cells in mice to show the system works in vivo. In addition, Chen says, physicians at Peking University First Hospital plan to use the method with cancer patients undergoing treatment with T cells engineered to fight their tumor. By expressing sortase A in the engineered T cells, the physicians could label tumor cells from patient blood samples that come into close proximity to the T cells. Sequencing RNA from those tumor cells could reveal gene signatures that correlate with resistance to immune therapies, Chen says.
The field is enjoying a number of new techniques for labeling proteins within living cells, and this evolved sortase is “a great new addition to the toolbox,” notes Peter V. Robinson, a chemist who creates tools for detecting proteins in living cells and who cofounded Enable Biosciences. He speculates, though, that you could get a biased signal if some cells have especially high or low numbers of glycines in their surface proteins.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter