Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Infectious disease
Covid-19
FDA outlines criteria for COVID-19 vaccine approvals
Clinical trials must prove a vaccine can prevent infections
by Lisa M. Jarvis
July 2, 2020
| A version of this story appeared in
Volume 98, Issue 26

The US Food and Drug Administration has given drug manufacturers a better idea of the data they will need for the agency to consider a COVID-19 vaccine ready for the public. The just-released guidelines come as more pharmaceutical companies provide early glimpses from vaccine trials.
According to the World Health Organization, 17 vaccines for COVID-19 had reached the clinic as of June 29, and another 132 are in early-stage development around the world. But even as they move through the pipeline at lightning speed, an open question has been what the FDA will want to see before it allows a vaccine on the market.
The document includes a few critical takeaways. The agency says it expects Phase III clinical studies to be gold standard: randomized, double blind, and placebo controlled—meaning neither doctor nor volunteer will know who is getting the vaccine or a placebo. Few of the hundreds of trials testing COVID-19 therapies—rather than vaccines—have followed that high standard, making it difficult to say with confidence which drugs work.
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
The FDA also explained what companies will need to prove a vaccine’s efficacy: late-stage data demonstrating that the vaccine can protect 50% of those receiving it from infection with SARS-CoV-2, the virus that causes COVID-19.
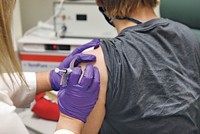
So far, early-stage COVID-19 vaccine studies have only looked for signs of worrisome side effects—few have been noted—and assessed whether vaccines are eliciting an immune response. In particular, companies like Moderna and CanSino have shown that people given their vaccines can generate neutralizing antibodies—the kind that prevent the virus from infecting cells.
Pfizer and BioNTech are the latest companies to offer neutralizing antibody data. On July 1, the partners gave a snippet of data from a Phase I/II trial of BNT162b1, one of four COVID-19 messenger RNA vaccines the pair is pursuing. The study included four arms, with volunteers receiving either a placebo, a single high dose, or one of two lower doses followed three weeks later by a booster shot.
Everyone who received the vaccine generated neutralizing antibodies—in some cases significantly more than in people who have recovered from the virus. The companies expect to begin a late-stage trial later this month of whichever vaccine looks best.
Conducting Phase III studies to hit the FDA’s efficacy mark—protection in 50% of vaccine recipients—will not be easy, especially if the pandemic is waning in areas where a trial is being conducted. The FDA says it is open to granting Emergency Use Authorizations for vaccines, a pathway that allows government stockpiling and public access amid the pandemic, but only after seeing full Phase III data.
The document dashes hopes of a vaccine reaching the US public this year. “The guidance makes clear that there are unlikely to be too many shortcuts for COVID vaccine clinical development compared to other vaccines,” SVB Leerink stock analyst Geoffrey C. Porges wrote in a note to investors.
While vaccine developers and the US government have proposed aggressive timelines for vaccines, Porges believes the more likely scenario is EUAs arriving in early 2021, with full approvals in 2022 or 2023 and widespread vaccination in 2023 or 2024.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter