Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Microbiome
Root chemicals help plant shape soil microbiome
The roots of Arabidopsis synthesize triterpenes that repel or attract certain soil microbes
by Alla Katsnelson, special to C&EN
May 13, 2019

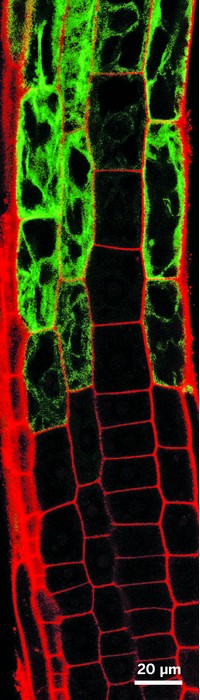
Plants may appear idle or passive to our eyes, but they are spectacularly busy, churning out a bewildering array of chemical signals that help them prosper in their environment. Some 20% of such chemicals are synthesized in a plant’s roots. Researchers have speculated that these organic molecules help regulate the distinctive microbial communities that surround roots in the soil, possibly selecting organisms that assist in plant growth or thwart pathogenic species. But scientists had yet to find direct evidence of such signaling.

Scientists now report that a small flowering mustard plant called Arabidopsis thaliana produces a swathe of previously-undiscovered chemicals belonging to a family of natural products called triterpenes (Science 2019, DOI: 10.1126/science.aau6389). About one-half of the microbes that are drawn to this plant’s roots are influenced by this chemical network, says Anne Osbourn, a plant biologist at the John Innes Center who co-led the work with Yang Bai, a plant biologist at the Chinese Academy of Science.
“The field has known that plant genotype influences root bacterial microbiome composition, but identifying the exact chemical mechanisms that drive these phenotypes is only beginning to be revealed,” says Sarah Lebeis, a plant microbiologist at the University of Tennessee, who was not involved in the work.

Osbourn’s team initially identified a group of genes that are expressed in the plant’s roots and that appeared to encode ecologically important metabolites. These genes, they found, encode three distinct metabolic pathways. One pathway synthesizes a triterpene the researchers called thalianin, another produces a compound they dubbed arabidin, and a third generates three triterpene fatty acid esters.
To investigate the function of these molecules, Bai, who studies how plants interact with soil microbes, planted Arabidopsis on a Beijing farm, along with mutant plants engineered to disrupt the synthesis of these five triterpenes. Then he and his lab used DNA sequencing to identify the microbes that had been attracted to the soil surrounding the different plants. “What he found was very surprising,” Osbourn says. “If Arabidopsis plants are blocked in their ability to make these root chemicals, they accumulate quite a different microbial community around the roots.”

Next, Osbourn’s lab synthesized some of the most abundant chemicals from the plants’ roots. They added these chemicals to microbes Bai’s team had isolated from near Arabidopsis roots, allowing the researchers to test the chemicals’ effects on the microbes directly in the lab. Some of the triterpenes inhibited the growth of certain microbes while other molecules promoted growth, mirroring the observations from the farm experiments. The researchers next plan to study the mechanism of the triterpene-microbe interactions in more detail to determine how exactly they affect growth.
The set of chemicals expressed by Arabidopsis are probably specific to the plant, almost like a fingerprint that attracts the specific line-up of microbes it needs, Osbourn says. By understanding how plants shape their microbial communities using such chemicals, researchers could extrapolate the findings to crop plants and perhaps devise ways to improve their health without fertilizers and pesticides.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter