Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Neuroscience
New tool could help researchers learn the language of the brain
Transistors decorated with DNA aptamers detect neurotransmitters
by Erika Gebel Berg
September 6, 2018
| A version of this story appeared in
Volume 96, Issue 36

The brain speaks in the language of neurotransmitters, but scientists are still far from fluent. Now, researchers have developed a device that can detect small molecules in the brain by turning the chemical signals into electrical ones (Science 2018, DOI: 10.1126/science.aao6750). The approach may yield clues about how neurotransmitters communicate fear, addiction, love, appetite, and all the other manifestations of our interior lives.
“I started working on this 20 years ago,” says Anne Andrews of the University of California, Los Angeles. “I’ve been trying to understand how serotonin encodes information about anxiety.” When she started her work, the existing methods for detecting neurotransmitter levels in the brain either were too slow or involved devices too large to observe the fluctuations in multiple neurotransmitters over time. But levels of neurotransmitters change quickly in small areas of the brain. So Andrews aimed to develop a method that was small, fast, and could detect different types of molecules.
Andrews found inspiration in a burgeoning technology: sensors based on field-effect transistors. Scientists had begun modifying field-effect transistors with protein receptors that could bind small biological molecules. When the receptor snares a target, the electric field around the transistor shifts, which can be read out as a change in current through the device.
Unfortunately, the approach is limited in biological environments like the brain by something called the Debye length—the farthest away a molecule can be from the transistor and still be detected. The greater a solution’s ionic strength, the shorter the Debye length because ions obscure the field effects created when the molecule binds to a receptor. Protein receptors are, in some cases, larger than the Debye length, making detecting the binding event difficult.
In a 2015 paper, Andrews worked with a different kind of receptor, a DNA aptamer, which is a nucleic acid chain that can be evolved to bind to a small molecule of choice (ACS Nano 2015, DOI: 10.1021/acsnano.5b01211). Compared to proteins, aptamers are tiny, so they might be compatible with short Debye lengths.
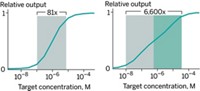
In the current study, Andrews and colleagues tested field-effect transistors with aptamers specific for dopamine, serotonin, glucose, and the zwitterionic lipid sphinghosine-1-phosphate. In solutions with ionic strengths similar to those inside the brain, the sensors’ current changed along with the concentration of target molecules but in different directions. The sensors detected increasing concentrations of sphinghosine-1-phosphate as increases in current. But increasing glucose and dopamine levels led to decreasing currents. According to the researchers, this suggests that the aptamers undergo different conformational rearrangements upon binding different molecules.
“This tool would help you understand how subsections of the brain interact and are modulated by different neurotransmitters,” says Jonathan Marvin, a protein engineer who develops tools for neuroscientists at Janelia Research Campus. “That would be a pretty powerful thing to learn.” Though he was disappointed the paper didn’t include a “proof of principle experiment in something alive.” He may get his wish soon, according to Andrews, who, after 20 years, is eager to start figuring out what all those neurotransmitters are saying in the brain.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter