Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Origins Of Life
Iron can catalyze metabolic reactions without enzymes
Findings suggest that the abundant metal might have played a key role in early biochemistry before enzymes evolved
by Alla Katsnelson, special to C&EN
May 1, 2019
| A version of this story appeared in
Volume 97, Issue 18

Enzymes catalyze the critical chemical reactions that help build and power living organisms. But chemists studying the origin of life on Earth face a vexing question: How did these metabolic reactions evolve before their enzymes existed?
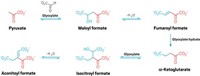
Take the Krebs cycle, for example. Also known as the citric acid cycle, it plays a central role in the metabolism of living things, breaking down carbon-containing molecules into carbon dioxide and producing energy in the form of adenosine triphosphate through a series of enzymatic reactions. The cycle, both its forward and reverse versions, is the only biochemical pathway to produce all five so-called universal metabolites: acetate, pyruvate, oxaloacetate, succinate, and α-ketoglutarate. “Every biological molecule you can make, its synthesis gets traced back to one of these five compounds on the Krebs cycle,” says Joseph Moran, a chemist at the University of Strasbourg.
Moran and his colleagues now report that a one-pot set of reactions involving ferrous iron and two simple molecules, pyruvate and glyoxylate, can generate all five universal metabolites (Nature 2019, DOI: 10.1038/s41586-019-1151-1). “We speculate that this pathway may have been the precursor to the Krebs cycle,” Moran says.
Until recently, most experimental work on the origin of life has focused on the interaction of reactive compounds, such as cyanide and formaldehyde. But that chemistry is fundamentally different from existing biochemistry, raising the question of how the latter evolved.
In the past five years, however, a handful of studies have begun to hint that the early Earth chemistry that produced life may not have had to reinvent itself. For example, a 2017 computational study mapped out a simplified molecular world that could still produce many biochemical reactions and molecules. Moran’s team noticed that in that primitive biochemistry, pyruvate and glyoxylate were lynchpin molecules. Last year, the group demonstrated how pyruvate could form without enzymes through a reaction involving CO2, iron, cobalt, and nickel.
Although the researchers still don’t know how glyoxylate could form under prebiotic conditions, they decided to see what kinds of reactions pyruvate and glyoxylate generate in an aqueous solution in the presence of different metals and under different experimental conditions, such as temperature and time. To the researchers’ surprise, the reaction network they observed with ferrous iron—the most abundant transition metal in the Earth’s crust—included all five of the universal metabolites. “It almost seems inevitable that if you ever had glyoxylate and pyruvate, they would have reacted in this way,” Moran says.
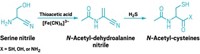
When the team added hydroxylamine and metallic iron into the mix, they produced four amino acids—glycine, alanine, aspartic acid, and glutamic acid—which are thought to be the first to arrive on the scene as biochemistry got its start. This observation suggests that cellular metabolism and the genetic code may have coevolved on Earth. “What’s interesting in our experiment is that we fairly selectively get only the compounds at the core of biochemistry,” Moran says.
The study identifies ferrous iron as the “one central electron donor—one central catalyst—that can actually do everything that we think is important in metabolism,” says Markus Ralser, a biochemist at the Francis Crick Institute. He thinks this paper may put to rest the old hypotheses of how biochemistry may have started on Earth. “Now, you can clearly see that it’s possible to make these molecules in the same fashion as cells do it,” he says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter