Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthetic Biology
Mirror-image polymerase makes key parts of mirror ribosome
Ongoing effort to create a reflection of life’s protein-making machinery could provide access to mirror molecules for diagnostics, therapeutics, and more
by Mark Peplow, special to C&EN
October 27, 2022

The ribosome is one of the most iconic machines in molecular biology—a complex structure that reads a recipe written in RNA to assemble amino acid building blocks into new proteins. Ting Zhu of Westlake University has long dreamed of creating a mirror-image version of the ribosome, a reflection of normal biochemistry that reads mirror RNA made from L-nucleic acids and makes mirror proteins from D-amino acids.
Zhu has now taken a significant step towards that goal by creating three crucial mirror RNA strands that will form the structural and catalytic core of a mirror ribosome, making up about two-thirds of its mass (Science 2022, DOI: 10.1126/science.abm0646). “Generating those large ribosomal RNAs is a key milestone towards building a mirror-image ribosome,” says Jonathan Sczepanski of Texas A&M University, who works with mirror RNA and mirror DNA and was not involved in the research. “It’s a really huge advance for the field of mirror-image biology.”
Zhu’s grand vision is to create a mirror-image version of the “central dogma” of molecular biology. In nature, DNA containing D-nucleic acids is replicated and transcribed into RNA, which the ribosome translates into proteins containing L-amino acids. “It’s the fundamental operating system of life as we know it,” Zhu says.
Recreating this sequence using chirally inverted components could give researchers a way to synthesize a plethora of mirror-image biomolecules for use in biosensors, medical therapies, and basic research. In Berlin, for example, TME Pharma (formerly Noxxon Pharma) is already developing short L-RNAs as cancer therapies. One key advantage is that these mirror molecules tend to resist digestion by ordinary enzymes. “From a biotechnology perspective, a lot of applications would be enabled by having a mirror-image ribosome,” Sczepanski says.
Although chemical synthesis techniques can make some mirror biomolecules, long strands of L-RNA—such as those needed for the core of the mirror ribosome—remain out of reach. Instead, Zhu’s team has synthesized a series of mirror polymerases that can replicate mirror DNA and transcribe it into mirror RNA. But their previous transcription polymerases haven’t been very efficient and only worked with single-stranded mirror DNA templates that are troublesome to prepare.
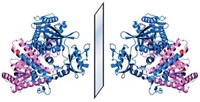
The researchers have now created a mirror version of T7 RNA polymerase, a highly efficient enzyme that uses more convenient double-stranded mirror DNA templates. Weighing in at 100 kDa, mirror T7 is far too large to synthesize in one go, so the team made it in three segments that could self-assemble into the full enzyme.
Then they created three mirror genes that coded for the crucial ribosomal RNA strands—known as 5S, 16S, and 23S—and used mirror T7 to transcribe them into RNA. The 23S strand contains 2,900 bases and is the largest piece of mirror RNA made to date.
The researchers still need to make many other ingredients before they can assemble a mirror ribosome, including dozens of ribosomal proteins and various other RNAs and proteins that help orchestrate the translation process. However, none are as large as the biggest mirror molecules already prepared in Zhu’s lab, suggesting that it should be possible to prepare them using the team’s existing techniques. Once all the parts are ready, Zhu hopes they can be coaxed to self-assemble into a complete mirror ribosome.
For now, mirror T7 itself could be a handy tool for those working on mirror biomolecules. As a proof of principle, Zhu used the polymerase to create an RNA construct known as a riboswitch, which produces a fluorescence signal when it binds guanine. Whereas natural-chirality RNA tends to be rapidly degraded by nucleases, the riboswitch sensor’s mirror chirality means it is left unscathed by normal biology. “The ability to make these things nuclease resistant and then introduce them into cells would be very powerful,” Sczepanski says.
CORRECTION:
This story was updated on Nov. 3, 2022, to correct a web production error. The handedness of the nucleic acids, amino acids, and RNAs are indicated with small-capital L and D and not with lowercase letters.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter