Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Toxicology
Hunting for the cause of mystery vaping illnesses
Little is known about the contents and toxicology of e-cigarette or THC products
by Britt E. Erickson
October 4, 2019
An updated version of this story was published on Oct. 10, 2019. Click here to read the new story.

No one knows for sure why more than 1,000 previously healthy individuals in the US, half under the age of 25, have developed serious lung injuries that require hospitalization and in many cases, mechanical ventilation and intensive care. Eighteen people have died from the mystery illness as of Oct. 3, according to the US Centers for Disease Control and Prevention. Health officials have ruled out an infectious agent. The only common link is that all of the victims have admitted to vaping nicotine or tetrahydrocannabinol (THC)—the psychoactive component in cannabis that gets you high—or both.
Federal, state, and local governments are scrambling to find answers to this emerging public health crisis. “No single e-cigarette or vaping product, brand, or specific substance has been definitively linked to the outbreak,” CDC’s principal deputy director Anne Schuchat told members of Congress at a Sept. 25 hearing.
The ongoing investigation is particularly challenging because it involves nearly all US states, Schuchat said. In addition, “the investigation is complicated by the diversity of the e-cigarette or vaping product marketplace, with a multitude of products, a wide array of ingredients, and the intersection with potentially illicit substances such as marijuana,” she noted. The bottom line is that “users do not know what is in their e-cigarette or vape solutions,” Schuchat said. And even if they did know, there’s little information available on inhalation toxicity.
The crisis, first reported by state health departments in July, has added urgency to an ongoing effort to stem the rise in e-cigarette use or vaping among youth in the US. The latest figures from the US National Youth Tobacco Survey suggest that 27.5% of high school students reported using e-cigarettes in the past 30 days. About 9% of all middle and high school students reported using cannabis in e-cigarettes.
Searching for a culprit
The nature of the lung injuries is one area of debate. A group from WakeMed Hospital in North Carolina reported in early September that lung injuries in five people resembled lipoid pneumonia, a condition in which fats or oils deposit in the lungs (Morb. Mortal. Wkly Rep., DOI: 10.15585/mmwr.mm6836e1). However, in a letter to the editor of the New England Journal of Medicine published on Oct. 2, Mayo Clinic and UnityPoint Health staff from Arizona, Florida, Illinois, and Minnesota report finding no evidence of lipoid pneumonia in lung tissue samples from 17 people with suspected vaping illness. The physicians instead point to lung inflammation “from one or more inhaled toxic substances” (DOI: 10.1056/NEJMc1913069).
While clinicians work on identifying and treating the lung injuries, others are actively looking for the cause. The US Food and Drug Administration is analyzing samples of vaping products consumed by those who have developed lung injuries. So far, the agency has received about 300 samples, acting commissioner Norman “Ned” Sharpless told members of Congress during the Sept. 25 hearing, held by the Subcommittee on Oversight and Investigations of the House Committee on Energy and Commerce.
FDA scientists at the agency’s Forensic Chemistry Center are analyzing the samples “using state-of-the-art methods to assess the presence of a broad range of chemicals, including nicotine; THC and other cannabinoids; opioids; cutting agents and other additives; pesticides, and toxins,” Sharpless testified. Such testing is limited, however, because many of the samples contain little to no liquid, he noted.
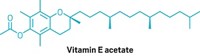
The FDA has analyzed about half of the 300 samples. Of those analyzed, about 70% contain THC, Sharpless said. About half of the products that contain THC also contain vitamin E acetate, he noted. The New York State Department of Health on Sept. 5 pointed to vitamin E acetate, an oil used as a nutritional supplement and as a diluent or thickening agent in THC vaping cartridges, as a possible cause of the lung problems. It is unclear whether vitamin E acetate is the cause of the lung injuries or is a marker that a product is adulterated, Sharpless said at the Sept. 25 hearing.

Nonetheless, cannabis testing labs are now starting to offer tests for vitamin E and vitamin E acetate in THC vaping cartridges. “It is not a standard test,” in the cannabis industry, says Amber Wise, science director at Medicine Creek Analytics, a Seattle-based cannabis testing lab. But vitamin E is used in many other products, such as lotions and nutraceuticals, “so analytical labs already have protocols for them,” she says. “It is a common thing you can test for in a food lab or product safety lab.”
Medicine Creek Analytics is testing for both vitamin E and vitamin E acetate, because it is unclear what additives are in THC vaping cartridges, she says. Wise suspects that “the acetate might be a breakdown product of vitamin E and not an actual compound that people are adding directly.”
Thickening agents are common in illicit THC cartridges, but they are rarely added to legal products where testing for potency is required, says Jeffrey Raber, cofounder and CEO of the Werc Shop, a California-based cannabis contract manufacturing and testing firm.
“THC concentrates are known to be thick and viscous when they are high potency,” Raber says. So when street dealers dilute illicit products with various agents to maximize profits, those products are typically less viscous. Consumers can visually detect the viscosity of the product by turning the cartridge upside down. If a bubble goes from the top to the bottom quickly, it usually means that the product has been cut with something, Raber says. Dealers mask that visual test by adding a thickening agent, so the bubble doesn’t move from the top to bottom as fast, and consumers think they are getting a high-potency product.
The illicit cannabis market “is out of control and concerning,” even in states like California where recreational cannabis is legal, Raber says. In California, the cannabis black market is estimated to be 3 to 4 times the size of the legal cannabis industry, he notes.
One source of the black market problem is that California requires testing of final finished cannabis products, Wise says. If a product fails the test, more often than not, it doesn’t get thrown away. Instead, it enters California’s black market and is then distributed to states where cannabis is illegal, she says.
The cannabis industry would like to see the black market problem addressed at the federal level. “If it is confirmed that Americans are being hurt because of unregulated, illicit market cannabis vape products, it is yet another reason for real, comprehensive federal cannabis reform that will allow the regulated, tested cannabis industry to displace illicit market actors,” says an Oct. 3 letter from the National Cannabis Industry Association to congressional leaders.
Although the majority of cases of lung injuries associated with e-cigarette or vaping products have involved the use of THC products, federal, state, and local authorities are not ruling out the possibility that nicotine e-cigarettes may also be playing a role. Joseph Allen and colleagues at Harvard T.H. Chan School of Public Health raised concerns in 2016 about three flavors—diacetyl, 2,3-pentanedione, and acetoin—widely found in e-cigarettes (Environ. Health Perspect., DOI: 10.1289/ehp.1510185). The flavors are used to give e-cigarette vapor a buttery, caramel, or fruity taste. They have been associated with severe respiratory disease in workers at microwave popcorn processing plants. The Harvard researchers reported finding at least one of the three chemicals in 47 of 51 e-cigarettes and liquids that they tested.
About 10–20% of the identified cases of lung injury from vaping so far involve patients who claim they were only using nicotine e-cigarettes. But that data is based on self-reporting, “and interviewees may be hesitant to share information about their use of illicit substances such as marijuana,” the CDC’s Schuchat acknowledged at the Sept. 25 hearing.
What is in the aerosols?
Determining what’s in either THC or nicotine vaping products is a key part of understanding what’s causing the lung injuries. However, what is in vaping cartridges is not the same as knowing what’s going into people’s lungs. “You can test the oil all you want, but at the end of the day, what are you inhaling?” Medicine Creek Analytics’ Wise asks. “That is the most important part to test in my opinion.”
Scientists at the CDC are analyzing the aerosols produced by some of the products consumed by patients with vaping illnesses, Schuchat testified. The analysis is complex because “users can modify the products, and the heating process can also influence the types and amounts of chemicals a user is exposed to,” she noted.
Cannabis testing labs are also investigating aerosols generated by various vaping products. Before the outbreak of lung illnesses hit the news, Medicine Creek Analytics ordered a vapor collection machine to test a filter device for a client to determine whether it removes pesticides, cannabinoids, and terpenes from cannabis vaping products, Wise says. The machine will be useful for understanding what is in the aerosol that people are vaping, she says.
And then there’s the question of which of the chemicals in aerosols might harm lung tissues. Little is known about the toxicity even of branded e-cigarettes, never mind THC products. The FDA claims that e-cigarette products are illegal because the agency has not approved any of them. But the agency has chosen to allow continued sales of e-cigarettes that were on the market before Aug. 8, 2016, the date when FDA’s regulation of e-cigarettes went into effect.
That is about to change, however. The FDA appears to be on the verge of requiring e-cigarette manufacturers to obtain agency approval for flavored e-cigarettes. And earlier this year, a US district court judge in Maryland ordered the FDA to begin enforcing the approval requirement for all e-cigarettes, not just those that contain flavors, by May 12, 2020. Manufacturers will need to show that “the product is appropriate for the protection of the public health,” the FDA regulation says. Meeting that standard will require demonstrating that the products do not cause harm.
Scientists at the Institute for In Vitro Sciences (IIVS) are working with tobacco companies to evaluate the inhalation toxicology of products in development or already on the market. They are using nonanimal, human cell–based testing platforms. The team is optimizing such tests for evaluating potential hazards associated with inhalable materials, including those from vaping devices, says Holger Behrsing, a principal scientist at IIVS.
“As part of our expansion of respiratory toxicology testing, we do utilize long-term pulmonary models and conduct repeat exposure studies to better recapitulate potential human exposures,” Behrsing says. “We believe such an approach using human, multicellular, three-dimensional respiratory test systems will better allow us to detect key events that may lead to more severe, adverse pulmonary conditions.”
Academic researchers are also tackling the problem. Earlier this year, for example, Allen and colleagues at Harvard reported the effects of the flavoring agents diacetyl and 2,3-pentanedione on human airway epithelial cells (Sci. Rep., DOI: 10.1038/s41598-018-37913-9). They found that both chemicals were associated with gene expression changes that could alter the production and function of cilia.
As for the cause of the current lung injury crisis, Wise believes people who vape a lot of nicotine e-cigarettes or THC cartridges probably have some existing lung damage. “And then they get a bad batch and it pushes them over the edge,” she says. The problem may also be related to the battery, the heating element, or the cartridge itself, she says, noting that many devices are cheap and not very reproducible. It is likely to be a combination of many factors, she says. “This situation really points to the need for oversight, regulation, and product safety testing.”
Advertisement
“I firmly believe that many aspects of the youth vaping epidemic could have been addressed if the FDA had moved forward with reviewing all e-cigarettes on the market when it first had the chance two years ago,” Rep. Frank Pallone Jr. (D-NJ), chair of the Energy and Commerce Committee, said at the Sept. 25 hearing. “These products have been marketed and targeted to kids without our knowledge of the full public health consequences.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter