Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Finance
Drugmakers post positive earnings for 2018
The industry’s outlook is sunny, with an emphasis on expensive new medicines despite pricing pressure
by Ryan Cross
February 15, 2019
| A version of this story appeared in
Volume 97, Issue 7

Most major drug companies reported sales and profit increases for 2018
Sources: Companies Note: Figures are percent change from 2017. a Based on adjusted net income after tax.
Drug company executives had a reassuring message for their stakeholders when reporting year-end earnings: we’re doing just fine.
Revenues and earnings increased for most major pharmaceutical firms in 2018. And during conference calls with investors, many companies forecast similar or increased revenues for 2019 as well.
The positive outlook comes at a time when the Trump administration has publicly shamed drug companies for price gouging. The US Department of Health and Human Services has proposed forcing companies to disclose drug prices during television ads, and President Donald J. Trump has even suggested that the US set limits on drug prices on the basis of what European countries pay.
Although tough talk from President Trump has shaken the industry before, in the most recent string of earnings conference calls, executives seemed unfazed by the political pressure. Instead, they emphasized drug pipelines stocked with increasingly complex and expensive medicines.
For many companies, this means investment in cancer therapies. Eli Lilly and Company, best known for its diabetes products, is expanding its reach in cancer through the $8 billion acquisition of Loxo Oncology. And GlaxoSmithKline, which sold most of its oncology portfolio to Novartis in 2015, is pivoting back to cancer with the recent $5 billion acquisition of Tesaro. Both deals indicate the high value of precision-oncology treatments, in which genetic testing is used to predict which people are most likely to benefit.
AstraZeneca CEO Pascal Soriot announced that his firm is restructuring to boost its emphasis on cancer. Its oncology R&D division will be led by José Baselga, who recently resigned as physician in chief at Memorial Sloan Kettering Cancer Center after accusations that he and others at the center failed to disclose financial conflicts of interest.
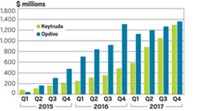
The king of oncology is the burgeoning field of immunotherapy and, in particular, drugs called checkpoint inhibitors, which are designed to help immune cells attack cancer. In 2018, sales of checkpoint inhibitors—which cost about $150,000 a year in the US—continued to soar. Merck & Co. posted sales of nearly $7.2 billion for its checkpoint inhibitor Keytruda, up 88% from 2017. And Bristol-Myers Squibb sold $6.7 billion worth of its checkpoint inhibitor Opdivo, up 36% from 2017.



During a call with investors, Hal Barron, GSK’s new head of R&D, said “the vast majority—actually, about 75% of the patients—either don’t benefit or will relapse” after treatment with checkpoint inhibitors. That suggests an opportunity for companies to improve upon the therapies.
Indeed, many firms are testing checkpoint inhibitors with other drugs in hundreds of so-called combination trials. In that vein, GSK recently announced a collaboration with Merck KGaA to develop a fusion protein that combines the backbone of a checkpoint inhibitor with another protein that sequesters an immunosuppressive molecule present around tumors. This therapy will be tested alone and also in combination with even more cancer therapies, Barron said.
Advertisement
Thirty-three of GSK’s 43 drug candidates in development target the immune system. The Merck KGaA partnership “is exactly the kind of thing that we want to continue to do,” said GSK CEO Emma Walmsley, who predicted “a meaningful uptick” in R&D spending this year.
Also part of the immunotherapy boom is investment in cancer cell therapies, though so far sales have been slow. Novartis posted only $28 million in sales from its CAR T-cell therapy Kymriah in 2018.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter